Innovation Awards Spark New Intramural Collaborations
Program Boosts Initiatives Supporting Researchers Across NIH

The NIH Director’s Challenge Innovation Awards Program funds projects that break down institutional barriers and bring together IRP scientists from disparate disciplines.
From Superbowl-winning football teams to comic book cohorts like The Avengers, combining the efforts of multiple talented individuals is a proven strategy for achieving remarkable results. It may come as no surprise, then, that the NIH’s Intramural Research Program (IRP) strongly encourages collaborations that breach the boundaries of its 24 Institutes and Centers. One example of these efforts is the Director’s Challenge Innovation Awards Program, which since 2009 has funded high-impact scientific projects that bring together researchers from across the IRP.
“We’re trying to create and encourage interactions among investigators from different NIH Institutes,” says Charles Dearolf, Ph.D., Director of Program Development and Support for the NIH’s Office of Intramural Research (OIR). “We think science moves better when multiple people with different perspectives are talking to each other.”
With the two years of funding provided by the Innovation Awards to help them get off the ground, the five initiatives selected this year will enhance IRP research in domains ranging from chronic pain to the immune system to blood-borne cancers. Read on for a round-up of these projects and their potential benefits for NIH scientists and patients around the world.
Zooming in on Critical Immune Cells
The human immune system is composed of multiple specialized components that contribute in different ways to the battle against dangerous pathogens like bacteria and viruses. Memory B cells, for instance, produce disease-fighting antibodies when they encounter specific molecules called antigens that pathogens carry. However, in people with immune deficiencies, B cells sometimes fail to respond to antigens on viruses or bacteria. Other times, the cells become active when they should not be, leading to allergies and autoimmune diseases in which the immune system attacks the body’s own tissues.
Consequently, learning more about B cells that respond, or fail to respond, to specific antigens could shed light on a wide array of illnesses. Unfortunately, this is easier said than done.
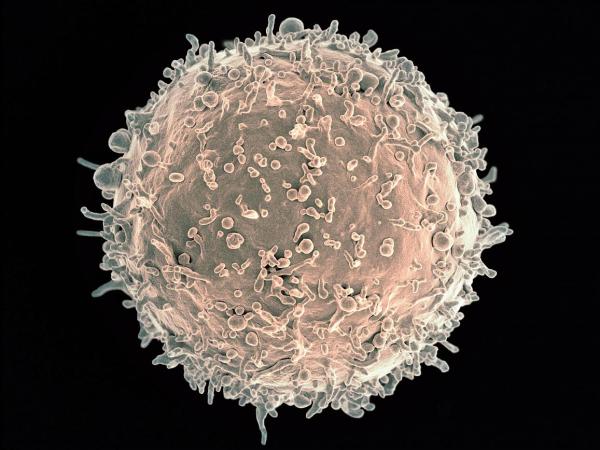
Colorized scanning electron micrograph of a B cell from a human donor.
“For many years, this was like looking for a needle in a haystack,” says IRP senior investigator Susan Moir, Ph.D., who studies the role of B cells in HIV. “Only a very small percentage of our B cells will make antibodies against a specific antigen, and to be able to pluck those out for analysis has been a very challenging task that doesn’t yield a lot of information.”
In an attempt to solve this problem, Dr. Moir and her collaborators are launching the B Cell Pilot Program (BCPP), which aims to assemble a set of advanced tools that IRP scientists can use to study specific varieties of B cells in great detail, as well as develop standardized methods for using those tools. Eventually, Dr. Moir and her colleagues hope to scale up the program into its own ‘core facility,’ a group of shared resources that any IRP researcher can use. The wide applicability of this communal toolkit, as well as the need for experts from disparate disciplines to come together to establish it, made the BCPP an ideal proposal for the Innovation Awards.
“This is a team effort,” says Dr. Moir. “My collaborators and I all come in with different expertise, but we’re also all interested in answering related questions, so it makes sense that we would bring our knowledge together to try to build something that could then be useful to the rest of the NIH community.”
Constructing Custom RNA
DNA has been the focus of so much research that it’s easy to forget its close cousin, RNA, also plays a vital role in how our bodies function. Studying the properties of specific RNA molecules and their contributions to health and disease requires scientists to accrue a sufficient quantity for their experiments. Rather than going through the tedious and difficult process of extracting RNA from cells, scientists often opt to synthesize the RNA molecules they wish to study from scratch in their labs. However, some RNA research also requires making specific chemical changes at particular locations in RNA molecules, and until recently it has been very technically challenging to construct long RNA molecules with these sorts of site-specific alterations in them.
“Think of it as the difference between covering an entire strand of spaghetti with sauce versus covering only one small bit one third of the way in from one side,” explains IRP senior investigator Adrian R. Ferré-D'Amaré, Ph.D., an expert on RNA.
Fortunately, in 2015, a group of scientists including Dr. Ferré-D'Amaré and led by IRP senior investigator Yun-Xing Wang, Ph.D., developed a new technique called position-selective labeling of RNA (PLOR) that allows scientists to construct large quantities of long RNA molecules that include customized chemical modifications. Now, through funding received from the Innovation Awards, Dr. Ferré-D'Amaré, Dr. Wang, and their collaborators plan to further hone the technique and figure out how to automate it, with the ultimate goal of establishing a ‘PLOR Core’ that would make the technology available to the entire IRP community.
“We think this is potentially transformative,” says Dr. Ferré-D'Amaré. “Labeling of RNA will revolutionize all aspects of RNA science and open up discoveries we cannot even dream of right now.”
Battling Blood Cancers
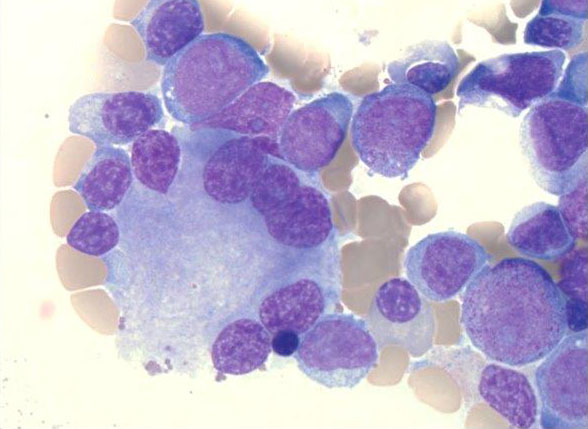
Abnormal stem cells in the bone marrow of a NIH patient with MDS/AML.
Cancer is an extremely diverse disease, so much so that even two patients with the same diagnosis might have cancers that differ in important ways. Cancers affecting blood cells, such as acute myeloid leukemia (AML) and myelodysplastic syndrome (MDS), are no exception to this rule, and are often even more difficult to treat than so-called ‘solid’ tumors because they are, by nature, spread out through the entire bloodstream rather than confined to one place. Consequently, the advent of precision medicine holds particular promise for combating these rare, highly fatal cancers.
“When people think about precision or personalized medicine, they’re thinking about treating the exact amount and type of cancer that a particular patient has at that point in time,” says AML researcher and IRP investigator Christopher Hourigan, M.D., D.Phil. “For each patient, we ideally would use deeper knowledge of their specific cancer to select the very best treatments.”
Producing those kinds of highly individualized therapies requires the ability to carefully scrutinize the characteristics of patients’ cancer cells, including the molecules present on them, the genetic changes that distinguish them from healthy cells, and the relationship between the two. Dr. Hourigan’s Innovation Awards proposal will establish a set of shared equipment and standardized methods that any IRP researcher studying AML or MDS can use to scrutinize these features. The analyses will not only reveal potential treatment targets, but they may also permit scientists to determine why certain patients in their clinical trials benefit from a therapy while others do not or why the same cancer reappears after a seemingly successful treatment in some people but not others. Moreover, by gathering data from many NIH clinical trials in one central repository, the initiative will enable IRP researchers to sift through the accumulated information for clues as to how to rid these patients of their cancers for good.
“We want to learn from everything that’s happening across the intramural program,” Dr. Hourigan explains. “These are rare yet diverse cancers, so for each 10- or 20-person clinical trial the chance of fully explaining these conditions is low, but if we start adding observations from across many trials, and we do it in a consistent way, we can make a real contribution to understanding and ultimately curing these terrible diseases.”
A One-Stop Shop for Pain Research
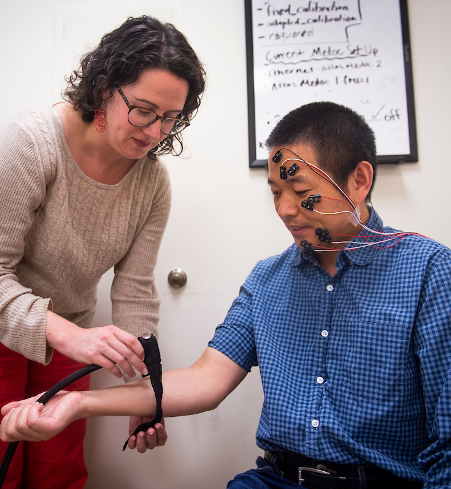
IRP investigator and pain researcher Lauren Atlas, Ph.D., applies equipment to a volunteer to measure his nonverbal responses to painful stimulation.
Not all Innovation Awards proposals pitch brand-new initiatives. A group of IRP scientists led by senior investigator Catherine Bushnell, Ph.D., will use funds from the program to expand the existing NIH Pain Research Center, which was established in 2019 to bring together IRP researchers studying all sorts of patient populations that experience pain as a part of their conditions, as well as NIH scientists working on pain treatments that do not rely on highly addictive opioid medications.
“Since most intramural researchers focus on a specific disease state, rather than on pain in general, a centralized Pain Research Center allows them to evaluate pain in their patients without having to recreate the necessary expertise and facilities,” says Dr. Bushnell. “Further, standardized pain testing across multiple disease states allows us to create a database to compare varied types of pain in order to better understand mechanisms and treatment options.”
Pain Research Center staff use a variety of standardized tools, such as questionnaires, sensory testing, imaging, and genetic sequencing, to glean information about pain in patients enrolled in many different NIH clinical trials, as well as healthy individuals for comparison. The Center already has two full-time staff members, testing space, and an array of equipment, but the Innovation Awards funding will enable the program to hire and train additional staff and expand its collection of instruments.
“I feel incredibly excited and grateful to receive these funds that will catalyze the NIH Pain Research Center,” Dr. Bushnell says. “We envision that with these funds, the Pain Center will very rapidly become a nexus for researchers to collaborate in deciphering mechanisms underlying pain associated with a multitude of diseases studied at NIH. It will transform intramural research into the neurobiological bases for chronic pain and related conditions, as well as the way we treat them.”
Scrutinizing Small Molecules
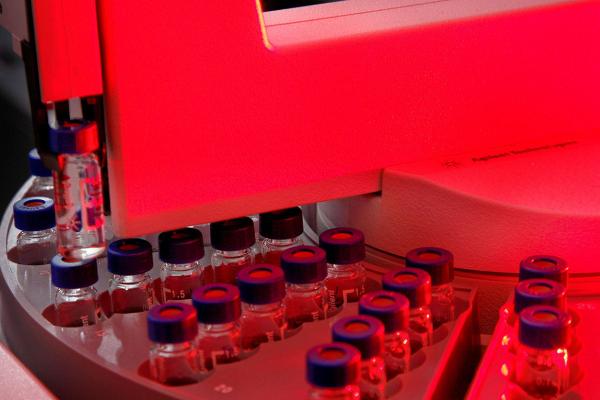
Image courtesy of Oak Ridge National Laboratory, US Department of Energy
Samples undergoing gas chromatography-mass spectrometry to analyze the small molecules present in them.
The molecules floating around inside our bodies come in a range of sizes, from large ‘macromolecules’ like the proteins that make up our muscles and organs to smaller compounds like sugars. The field of metabolomics focuses on these small molecules, with the sub-discipline of lipidomics particularly dedicated to fat-related molecules called lipids.
Learning about small molecules can lead to advances in treating and even predicting numerous diseases, and because metabolomics involves the study of so many different substances, it crosses paths with a huge number of research domains. However, there has been little work done to bring together disparate IRP researchers whose work involves metabolomics. A group of IRP scientists led by senior investigator Michael Fessler. M.D., hopes to remedy this by using Innovation Awards funding to create a Trans-NIH Metabolomics and Lipidomics Consortium.
“Institutions outside NIH have succeeded in achieving coordinated advances in standardized methodology over the past several years by virtue of the NIH Common Fund,” says Dr. Fessler, who studies the role of cholesterol-based molecules in the immune system. “Here at NIH, this coordination across groups using metabolomics has not existed. Hence, there has been no standardization of methods and little shared institutional knowledge or economy of scale. The Consortium will serve to build these bridges across the IRP.”
The Consortium has already organized an advisory committee made up of representatives from multiple NIH institutes and taken a census of the equipment and expertise available at NIH for metabolomics work. It is now turning its focus to constructing reference libraries of small molecules and a website where researchers can access standardized methods. The program will also explore the possibility of creating a centralized metabolomics core facility to which any IRP researcher can submit samples for analysis. By breaking down the walls between NIH scientists with different specialties, Dr. Fessler hopes the Consortium will dramatically accelerate research on the many small molecules that keep us healthy or drive disease.
“This is a very exciting opportunity to build a sustainable program at NIH that fills a critical unmet research need,” Dr. Fessler says.
As our scientific knowledge grows, researchers are increasingly identifying important overlaps in their domains of expertise. In the IRP, the Innovation Awards are expanding the resources and kick-starting the interdisciplinary collaborations that will be needed to further unravel the mysteries of our biology.
Subscribe to our weekly newsletter to stay up-to-date on the latest breakthroughs in the NIH Intramural Research Program.
Related Blog Posts
This page was last updated on Tuesday, May 23, 2023
