Experimental Treatment Wards Off Blinding Autoimmune Disease
Drug Candidate Calms Overzealous Immune Response in the Eye
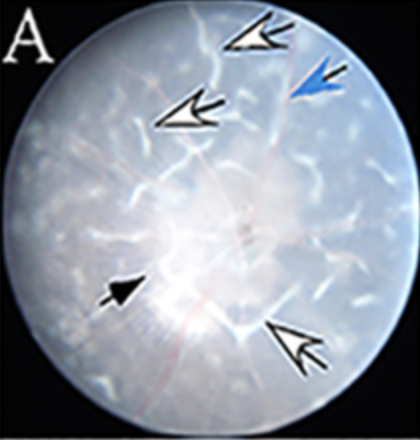
Hyperactive immune cells in the eye cause damage (indicated by arrows) and lead to a potentially blinding ailment called autoimmune uveitis. IRP scientists tamed those cells in mice using a drug called AS101. Photo courtesy of Charles Egwuagu
Our immune cells don’t like strangers and attack many organisms and substances that they have never seen before, including harmless ones. In autoimmune diseases, this reaction gets out of hand and our own cells are caught in the crossfire. IRP scientists have found that a new therapeutic compound can curb this sort of autoimmune carnage in the eye.1
Vision relies on a variety of chemicals, and because these substances are normally confined to the eye, the immune system doesn’t recognize them as a normal part of the body. Consequently, when immune cells somehow encounter these eye-specific proteins, or molecules that resemble them, the cells declare war on those proteins throughout the body, including in the eye where they rightfully belong. The resulting damage to the eye’s light-sensing retina, known as autoimmune uveitis, is one of the major causes of preventable blindness.
“By and large, we don’t know where the trigger comes from that causes the body to attack its own eye,” says IRP senior investigator Rachel Caspi, Ph.D., the new study’s senior author. “It’s probably something that’s sufficiently similar to a retinal protein that it fools the immune system into thinking that the retinal proteins are also the enemy.”
Current treatments for autoimmune uveitis, such as steroids, have serious side effects. As a result, Dr. Caspi’s team turned to a new compound called AS101 that is noted for its minimal side effects and works by inhibiting molecules called integrins that play a role in uveitis. The drug has also helped patients in trials for a wide range of illnesses, including human papilloma virus (HPV), the inflammatory skin ailment psoriasis, and the vision-destroying condition known as age-related macular degeneration.
“Anything that can reduce the amount of steroids that a patient will remain stable on is already good,” Dr. Caspi says. “AS101 is very non-toxic, so there seems to be nothing to lose. It’s a drug that, logically, should affect at least some of the processes that we believe are involved in uveitis by virtue of its biochemical activity.”
The researchers began by giving healthy mice a daily dose of AS101 for two weeks. Just one week into the treatment regimen, the mice had many more immune cells called regulatory T cells, which are critical to tamping down the immune response and preventing other immune cells from attacking the body’s tissues. Moreover, in mouse models of autoimmune uveitis, the eyes and lymph nodes of animals treated with the drug contained fewer of the type of immune cells that attack foreign substances, known as helper or effector T cells. Consequently, the treated mice had markedly reduced damage to their eyes. The AS101-treated animals also had more regulatory T cells in their spleens, an important reservoir for immune cells in the body.
Further experiments revealed that AS101 can encourage immature immune cells to develop into regulatory T cells and inhibit cellular processes that change immune cells into effector T cells. The drug also showed the ability to calm the damaging behavior of certain varieties of effector T cells. Together, these findings suggest that AS101 curbs autoimmune uveitis by increasing the ratio of beneficial to harmful immune cells and by making the harmful cells less active.
Now that AS101 has proven beneficial in animal models of autoimmune uveitis, Dr. Caspi would like to see the compound tested in clinical trials for the condition. Her research group also plans to continue studying the mechanisms by which AS101 alters the immune system.
“But as far as testing clinical efficacy, we don’t necessarily need to wait until we know the mechanisms in great detail,” Dr. Caspi says. “Since AS101 has a clinical history of being nontoxic and has already been shown to be efficacious in clinical trials for a variety of inflammatory diseases — both autoimmune diseases and conditions very different from uveitis — it makes sense to try it.”
Subscribe to our weekly newsletter to stay up-to-date on the latest breakthroughs in the NIH Intramural Research Program.
References:
[1] AS101 ameliorates experimental autoimmune uveitis by regulating Th1 and Th17 responses and inducing Treg cells. Bing SJ, Shemesh I, Chong WP, Horai R, Jittayasothorn Y, Silver PB, Sredni B, Caspi RR. J Autoimmun. 2019 Mar 7. pii: S0896-8411(19)30020-4. doi: 10.1016/j.jaut.2019.02.006. [Epub ahead of print]
Related Blog Posts
This page was last updated on Wednesday, May 24, 2023
