Research Briefs
Read about NIH scientific advances and discoveries by intramural scientists: mechanical properties of cancer cells influence their aggressiveness; proteins that restore hearing in zebrafish; medication to treat alcohol-use disorder; novel imaging reveals details of a rare eye disease; compounds that may reverse neurological disease; and vaccines to prevent reinfection with hepatitis C.
NCI: MECHANICAL PROPERTIES OF CANCER CELLS GIVE INSIGHT INTO METASTASIS

CREDIT: KANDICE TANNER, NCI-CCR
NCI researchers used a technique called Brillouin microscopy to explore how cancer cells’ biomechanical properties influence their growth and aggressiveness. Shown: A spheroid of breast cancer cells grown in a three-dimensional matrix; Brillouin microscopy reveals each cell’s stiffness (darker red cells are stiffer).
What makes some cancers spread aggressively while others remain harmless for years or decades? One theory is that cancer cells tend to be pliable and can adopt different mechanical properties that are thought to help them slip through tissues and migrate to distant sites in the body. In a recent study, NCI scientists showed how noninvasive techniques can be used to explore how a cancer cell’s mechanical properties are shaped by external cues from its environment.
According to lead author Kandice Tanner, cells’ mechanical properties—their stiffness and viscosity, for example—have such a profound influence on their behavior that changes to these properties are likely key to cancer’s growth and progression.
The research team assessed the viscoelasticity of cancer cells growing in complex three-dimensional structures with a technique called Brillouin microscopy, which tracks the way light scatters when it hits materials. Paired with a method developed in Tanner’s lab called optical tweezer microrheology, which uses microbeads to apply precise forces to the cells, the researchers quantified the mechanical properties at both cellular and subcellular levels.
Cells can alter their mechanical properties in response to their environment. “We’re really probing what a cell senses in the context of a tissue, and we’re doing it under different microenvironmental cues that mimic what the cells would see in a living animal,” said Tanner. She hopes that biomechanical information might one day help clinicians predict whether a patient’s tumor is likely to metastasize and if it does, where metastatic tumors are most likely to emerge. (NIH authors: M. Nikolic and K. Tanner, Biophy J 121:3586-3599, 2022; DOI:10.1016/j.bpj.2022.09.002)
NHGRI, NLM, NIAID: UNIQUE SET OF PROTEINS RESTORES HEARING IN ZEBRAFISH
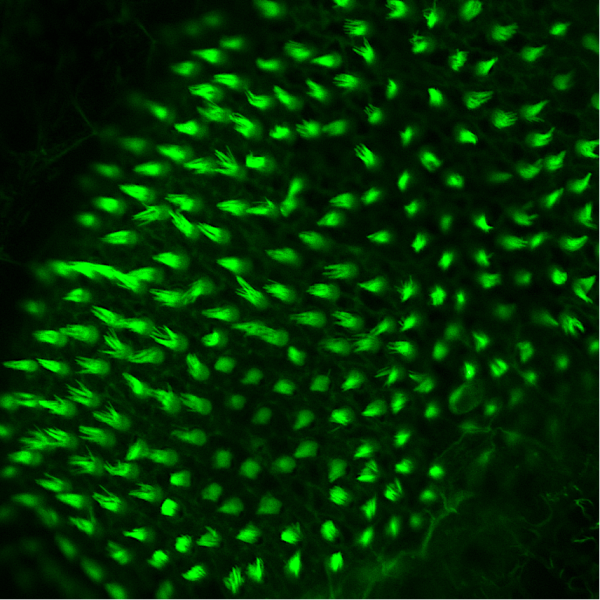
CREDIT: ERIN JIMENEZ, NHGRI
Confocal image of adult zebrafish hair cells (green) in the auditory organ of the inner ear. NIH researchers identified a mechanism for hair-cell regeneration in zebrafish that could one day translate to a treatment to correct hearing loss in humans.
Through aging and trauma, hearing loss in humans is often caused by the loss of hearing receptors in the inner ear known as hair cells. Humans can’t regenerate hair cells, but some animals, such as zebrafish (Danio rerio), can. A genomic study led by NHGRI researchers has identified a mechanism for cell regeneration in zebrafish that could one day translate to humans.
Using a combination of genomic techniques and computational-based machine learning, the investigators found that the regeneration of hair cells in zebrafish relied on a network of transcription factors, which are proteins that can switch genes on and off.
Two families of transcription factors were identified that work together to activate hair-cell regeneration in zebrafish, called Sox and Six. When hair cells die in zebrafish, nearby support cells start changing to become new hair cells in a process called transdifferentiation. Like stem cells, support cells can become other cell types.
First, the Sox transcription factors initiate the regeneration response in surrounding support cells. Next, both Sox and Six cooperate to turn those support cells into hair cells. “This group of zebrafish transcription factors might become a biological target that may lead to the development of novel therapy to treat hearing loss in humans,” said lead author Erin Jimenez. (NIH authors: E. Jimenez, C.C. Slevin, W. Song, S.C. Frederickson, D. Gildea, W. Wu, A.G. Elkahloun, I. Ovcharenko, and S.M. Burgess, Cell Genomics 2:100178, 2022; DOI:10.1016/j.xgen.2022.100170)
NEI, NIAID, NINDS: NOVEL IMAGING APPROACH REVEALS DETAILS ABOUT RARE EYE DISEASE

CREDIT: JOHNNY TAM, NEI
Retinal pigment epithelium cells (RPE) in a male participant with choroideremia, showing that enlarged RPE cells can be detected using a multimodal imaging approach (see circled examples).
For the first time, NEI scientists have shown how cells across different tissue layers in the living human eye are affected in people with choroideremia, a rare genetic disorder that leads to blindness.
Lead author Johnny Tam combined adaptive optics (a technology that enhances imaging resolution) with indocyanine green dye to view live cells in the retina, including light-sensing photoreceptors, retinal pigment epithelium (RPE), and choroidal blood vessels. His team was able to visualize how choroideremia disrupts these tissues, providing information that could help design effective treatments for this and other diseases. The retina’s RPE is a layer of pigmented cells essential to the nourishment and survival of photoreceptors.
“One major finding of our study was that the RPE cells are dramatically enlarged in males and females with choroideremia,” said Tam. His team also found that enlarged RPE cells can be detected even when using only a commercially available scanning laser ophthalmoscope along with indocyanine green dye. Tam noted that using an existing tool in the clinic could help clinicians identify which patients would benefit the most from therapeutic interventions. (NIH authors: N. Aguilera, T. Liu, A.J. Bower, J. Li, S. Abouassali, R. Lu, J. Giannini, M. Pfau, C. Bender, M.G. Smelkinson, A. Naik, B. Guan, O. Schwartz, A. Volkov, D. Maric, R. Fariss, R.B. Hufnagel, B.G. Jeffrey, B.P. Brooks, W.M. Zein, L.A. Huryn, and J. Tam, Commun Biol 5:article number 893, 2022; DOI:10.1038/s42003-022-03842-7)
NIDA, NIAAA: POTENTIAL NEW MEDICATION TO TREAT ALCOHOL-USE DISORDER
One size does not fit all when it comes to treating alcohol- and substance-use disorders. Currently there are three medications approved for alcohol-use disorder in the United States, but new research by collaborative teams at NIDA, NIAAA, and the Yale School of Medicine (New Haven, Connecticut) shows promising results for repurposing spironolactone as another potential pharmacotherapy for the disorder.
Spironolactone is used to treat cardiovascular conditions such as hypertension and heart problems. It works by inhibiting hormone receptors found throughout the body that regulate electrolyte balance. Preclinical and clinical research has suggested that those hormones and receptors might play a role in alcohol use, and the mechanism of action of spironolactone is actively being investigated.
Laboratory studies led by NIDA co-senior author Leandro Vendruscolo discovered that administering spironolactone decreased bingelike drinking in male and female mice. The drug also reduced ethanol self-administration in alcohol-dependent male and female rats without causing movement problems or affecting water intake throughout the studies.
Concurrently, researchers at Yale led retrospective studies on a large cohort of patients from the United States Veterans Affairs health care system. The scientists found that patients who were prescribed spironolactone for cardiovascular conditions had a marked reduction in self-reported alcohol consumption. According to the authors, the findings support the need for randomized controlled trials of spironolactone in people with alcohol-use disorder. (NIH authors: M. Farokhnia, V. Chuong, M.A. McGinn, S.K. Elvig, E.A. Douglass, L.A. Gonzalez, J.E. Sanfilippo, R.C.N. Marchette, B.J. Tunstall, G.F. Koob, L. Leggio, and L.F. Vendruscolo, Mol Psychiatry 2022; DOI:10.1038/s41380-022-01736-y)
NIDDK, NIAID, ORS: CELLS THAT REMEMBER HEPATITIS C VIRUS PROVIDE OPPORTUNITY FOR VACCINE DEVELOPMENT
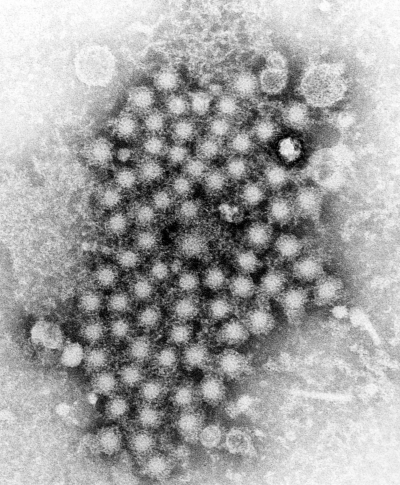
CREDIT: E.H. COOK JR, CDC
The findings from an NIH-led study suggest that vaccines could be developed to protect people from being reinfected with the hepatitis C virus. Shown: A transmission electron microscopic image of hepatitis virus particles.
Findings from a study led by NIDDK researchers suggest that vaccines could be developed to protect people from being reinfected with the hepatitis C virus (HCV). The researchers calculated the duration and quality of the natural antibody response against hepatitis C by following HCV-cured patients for up to 18 years. The natural antibody response declined rapidly, with a half-life of about five to seven years. However, some cells continue to remember the virus for decades after the virus was cured. In response to an antigen on the surface of a virus, these long-lived memory B cells are activated and multiply rapidly, differentiating into plasma cells that secrete protective antibodies. The cells’ persistence and ability to secrete antibodies suggest that they could be pertinent to the development of future HCV vaccines. (NIH authors: A. Nishio, S. Hasan, H. Park, N. Park, L. Kardava, P. Juneau, S. Moir, M.G. Ghany, and B. Rehermann, Nat Commun 13:5446, 2022; DOI:10.1038/s41467-022-33035-z)
NCATS: NOVEL MOLECULAR COMPOUNDS MAY REVERSE NEUROLOGICAL DISEASE
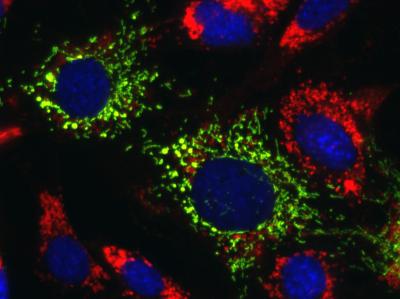
CREDIT: YIANNIS IOANNOU LAB, DEPARTMENT OF GENETICS AND GENOMIC SCIENCES, ICAHN SCHOOL OF MEDICINE AT MOUNT SINAI
NCATS researchers and colleagues at the Icahn School of Medicine at Mount Sinai reversed the effects of several life-threatening, inherited neurodegenerative diseases called lysosomal storage disorders (LSDs). Shown: Lipid-filled lysosomes (red) from Niemann-Pick disease type C1 (a type of LSD) are normalized when mitochondrial TRAP1 (green, top left and center) is activated.
NCATS researchers and their colleagues at the Icahn School of Medicine at Mount Sinai (New York) have identified small-molecule compounds that reversed the effects of several life-threatening neurodegenerative diseases called lysosomal storage disorders (LSDs) in patient cells and mice. LSDs are caused by inherited genetic defects in which a cell’s lysosomes stop breaking down and recycling fats, sugars, and proteins, which can accumulate in the liver and brain. This accumulation eventually causes the energy-producing mitochondria to malfunction. In a recent study, the scientists uncovered new compounds that restored mitochondrial function by activating the tumor necrosis factor receptor-associated protein 1 (TRAP1).
The Mount Sinai researchers first developed assays to identify compounds that increased the expression of Rab9, an enzyme associated with improved lipid storage in a type of LSD known as Niemann-Pick disease type C1 (NPC1). A NCATS team led by Juan Marugan then used the test with high-throughput screening facilities to sift through thousands of compounds and determine which ones worked best. Active molecules were chemically optimized and further evaluated on NPC1 cells. The investigators discovered that some compounds activated TRAP1 and made the mitochondria work properly again, and also restarted the lysosome’s fat-recycling ability.
Additionally, increasing TRAP1 activity in cells from patients with other LSDs also corrected those cell’s respective conditions.
Similar dysfunctions in cell recycling and mitochondrial health occur in other neurodegenerative disorders, such as Parkinson disease, amyotrophic lateral sclerosis, and Alzheimer disease. “We believe that this approach could have therapeutic benefits for more mainstream disorders, as well,” said Marugan. (NIH authors: R. Calvo, S. Patnaik, R. Mull, P. Dranchack, A. Wang, X. Xu, E. Hughes, N. Southall, M. Ferrer, and J.J. Marugan, iScience 25:104941, 2022; DOI:10.1016/j.isci.2022.104941)
This page was last updated on Wednesday, November 9, 2022
