Inflammation Contributes to Cancer-Related Fatigue
Mouse Study Suggests Approach to Combat Patients’ Debilitating Tiredness

New IRP research suggests that the intense feelings of exhaustion experienced by many cancer patients could be caused in part by inflammation in their bodies triggered by radiation therapy.
The human body is like any delicate ecosystem — disrupting just one part of it can have unexpected, widespread repercussions. Cancer patients know this well, not just because a tumor confined to one organ can cause a range of symptoms, but also because radiation treatment aimed specifically at the tumor sometimes leaves patients feeling utterly exhausted. New IRP research suggests that an inflammatory response to targeted radiation therapy is responsible for this common side effect of the treatment.1
One might expect cancer chemotherapy, which introduces potent chemicals into the blood, and whole-body radiation treatment to cause severe side effects, but even when radiation is targeted to just one organ, it can cause debilitating feelings of tiredness, referred to as ‘cancer-related fatigue.’ IRP senior investigator Leorey Saligan, Ph.D., R.N., for example, has observed that patients with prostate cancer involved in his studies don’t initially mention feeling particularly tired, but all of them develop significant fatigue when they begin receiving radiation targeted to the prostate. What’s more, up to 40 percent of them continue to experience chronic fatigue for a year or two after their treatment ends.
“The symptom of fatigue has always been used synonymously with being tired, so people think it can be dismissed or easily relieved by taking a nap or taking some time off, but actually it isn’t so simple,” Dr. Saligan says. “This is not just being tired where you can take a nap and it gets better. One of these patients mentioned that it’s hard for them to even open their eyes. That really affects everything: they’re less productive, they’re irritated because they can’t do as much, and most of the time it affects their social roles — not just their work but being a parent or a caregiver.”
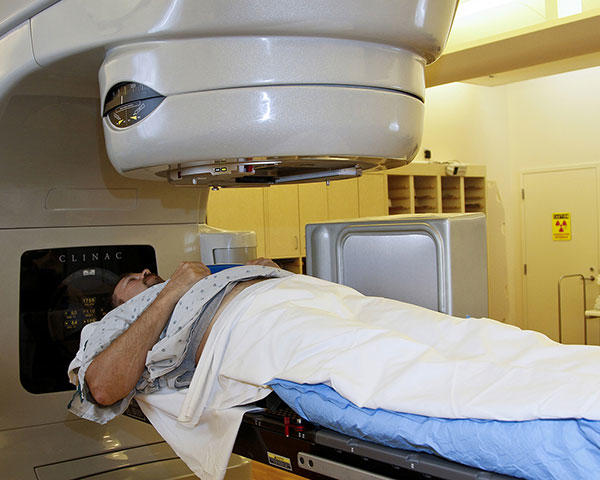
A patient receiving radiation treatment for cancer.
Since the cancer itself did not seem to be causing fatigue in Dr. Saligan’s patients, he began to investigate how their radiation treatment was triggering feelings of exhaustion despite only being aimed at the site of their tumors. Delving into the scientific literature, Dr. Saligan learned that many studies had identified signs of significant inflammation in the bodies of patients with cancer-related fatigue. However, nobody had established that this inflammation was actually causing the fatigue. In a new study, his team aimed to establish that causal link by examining how interventions that reduce inflammation affect physical activity in a mouse model of cancer-related fatigue that his lab had previously developed.2
In the new study, just like in prior experiments done in Dr. Saligan’s lab, healthy male mice that received radiation targeted to the pelvis spent significantly less time running on their wheels than control mice not exposed to radiation. The scientists interpreted this as a sign that the animals, like their human counterparts, were feeling less energetic after the treatment. However, mice that were given minocycline, an antibiotic that also reduces inflammation, showed a noticeably smaller decrease in their wheel running than mice not given the antibiotic, though they still ran much less than mice that were not exposed to radiation. Similarly, after pelvic radiation treatment, mice lacking a gene involved in the body’s inflammatory response, called MyD88, ran more on their wheels than genetically normal mice.
“Minocycline is a broad-spectrum antibiotic, and when you’re on an antibiotic there are a lot of side effects, particularly related to your gut,” Dr. Saligan explains. “We know that the gut and the brain have direct access to each other, and changes in the gut can affect behavior. By using mice without MyD88, we avoided any changes in the gut that may affect the fatigue behavior.”
More From the IRP
Research in Action
Energized by Fatigue
Changes in immune signaling molecules called cytokines also pointed to inflammation as a contributor to the animals’ fatigue-like behavior. Specifically, mice given pelvic radiation had higher levels of a pro-inflammatory cytokine called IL-6 in their blood, but this increase was blunted in animals given minocycline. On the other hand, Dr. Saligan’s team failed to find significant changes in cytokine levels throughout the animals’ brains after exposure to radiation.
Of course, just because the IRP researchers could not detect altered cytokine levels in the entire brain doesn’t mean there were not isolated changes in the amount of cytokines in specific brain regions. As a result, Dr. Saligan’s team plans to examine whether introducing IL-6 or other cytokines into specific parts of a mouse’s brain can induce fatigue-like behavior. The group also hopes to develop an animal model that shows longer-lasting post-radiation changes in behavior more akin to the lingering fatigue experienced by some patients participating in Dr. Saligan’s studies.

Dr. Saligan works with a participant in one of his studies.
Despite new the study’s limitations, Dr. Saligan sees it as an important “stepping stone” along the path to treatments that alleviate cancer-related fatigue.
“This study fills a gap in the literature,” Dr. Saligan explains. “Past studies have only found associations between inflammation and fatigue, so I think this is a big first step to really show some causality between fatigue and inflammation. We’re hoping that with these initial results that show a causal relationship, we can provide avenues to address the role of inflammation in cancer-related symptoms such as fatigue.”
Subscribe to our weekly newsletter to stay up-to-date on the latest breakthroughs in the NIH Intramural Research Program.
References:
[1] Inflammation plays a causal role in fatigue-like behavior induced by pelvic irradiation in mice. Wolff BS, Alshawi SA, Feng LR, Juneau PL, Saligan LN. Brain Behav Immun Health. 2021 May 19;15:100264. doi: 10.1016/j.bbih.2021.100264.
[2] A Mouse Model of Fatigue Induced by Peripheral Irradiation. Wolff BS, Renner MA, Springer DA, Saligan LN. J Vis Exp. 2017 Mar 17;(121):55145. doi: 10/3791/55145.
Related Blog Posts
This page was last updated on Tuesday, May 23, 2023

