That Record-breaking Sprint to Create a COVID-19 Vaccine
Kizzmekia Corbett and Barney Graham Recognized for Leading IRP Vaccine Research

CREDIT: CHIA-CHI CHARLIE CHANG
Kizzmekia Corbett (left) and Barney Graham (right) were named finalists for the 2021 Samuel J. Heyman Service to America Medals for leading NIH’s COVID-19 vaccine development effort.
At the end of 2019, most people were looking forward to an exciting 2020, a new decade starting with those magic numbers, 20-20, that denote a sharpness of vision. There would be the Summer Olympics in Japan and the U.S. presidential election. Meanwhile, intramural scientists at the National Institute of Allergy and Infectious Diseases’ Vaccine Research Center (VRC) were designing vaccines for several coronaviruses using a promising, new platform based on messenger RNA (mRNA).
Everything changed on a Saturday morning in early January. Chinese scientists had isolated a new coronavirus that was causing a serious epidemic in China’s Wuhan province and released its genetic sequence to the scientific community around the world. Barney Graham, director of the VRC’s Viral Pathogenesis Laboratory (VPL), and VRC research fellow Kizzmekia Corbett dropped everything and began using this mRNA platform to develop a vaccine for the illness that would become known as COVID-19.
“Dr. Corbett was directing a team doing coronavirus work, and we had relationships with three or four really good academic collaborators and had been having monthly conference calls for years,” Graham said. “We also had our industry collaborators [at Moderna], and we had a strategy and all the technology, so we were ready to go.”

CREDIT: NIH
Barney Graham’s lab is located in NIH’s Dale and Betty Bumpers Vaccine Research Center, also known as Building 40.
Graham and Corbett quickly mapped out a plan to begin experiments, assigned roles to team members, and went to work designing an antigen—in this case, a copy of the spike protein found on the surface of the COVID-19 virus, which it uses to infect cells. This antigen molecule, when provided via a vaccine, would trick the body into forming a defensive arsenal against future infection. After years of prior work to unlock the mysteries of coronaviruses and, with their partners at Moderna, perfect a method to coax the body to manufacture antigens via mRNA, they were prepared. Less than 48 hours after the release of the novel coronavirus’s genome, the team had designed the protein that their candidate COVID-19 vaccine would use to teach the immune system to fend off the virus. Sixty-five days later, the VRC began clinical trials in collaboration with Moderna and clinical investigators from NIH’s Division of Microbiology and Infectious Diseases. Ultimately, the vaccine received emergency use authorization (EUA) from the FDA on December 18, 2020, just one week after a similar vaccine developed by Pfizer was granted an EUA.
In recognition of this groundbreaking success in developing a life-saving COVID-19 vaccine in record time, Graham and Corbett were named finalists for the 2021 Samuel J. Heyman Service to America Medals, also known as the “Sammies.” Often referred to as the “Oscars of government service,” the Sammies honor exceptional work by government employees.
Although Graham and Corbett have captured the public’s attention, they both stressed that there were dozens of other researchers at the VRC and elsewhere that made the NIH–Moderna vaccine a reality.
A Long Road and a Fast Finish
While creation of the specific vaccine for COVID-19 was surprisingly rapid, Graham and Corbett, along with fellow researchers in their field, had been laying the groundwork for decades. As a young chief resident at Vanderbilt University School of Medicine (Nashville, Tennessee), Graham began studying respiratory syncytial virus (RSV), an infectious disease that can be fatal in children. A vaccine made with the inactivated virus had been tested in the 1960s with tragic results—not only did it not work, but the disease worsened in children who received the vaccine.
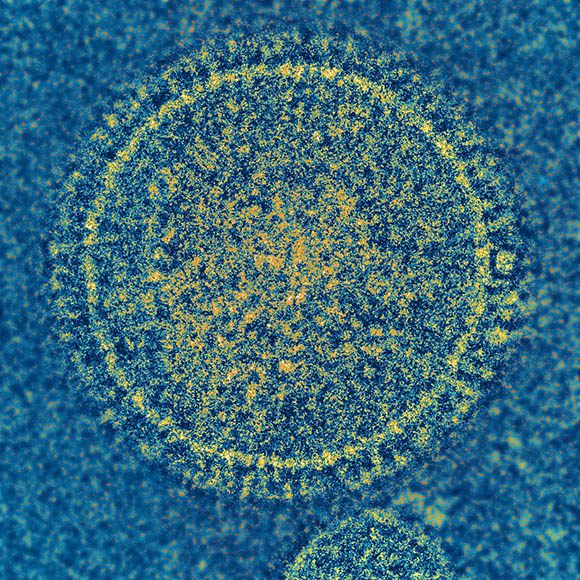
CREDIT: NIAID
Graham’s research on the respiratory syncytial virus, pictured here, left him well-situated to transition to studying coronaviruses like the one that causes COVID-19.
Eventually, Graham discovered that the original vaccine failed for two major reasons. First, the vaccine induced a response from immune cells called T cells that was more like an allergic response, making a lot of mucus and not effectively clearing the virus from the body. Second, inactivating the virus caused it to change shape to the form it takes once it has already infected and fused with a cell. As a result, the vaccine caused the body to release antibodies that could bind to the virus but could not block infection, making it ineffective. These findings set Graham on a path to create vaccines that could emulate the prefusion form of the virus.
He continued the RSV project in 2000 when he was recruited to NIH to help create the VRC. Eventually, he and Jason McLellan, a postdoctoral fellow in the lab of senior investigator Peter Kwong isolated and created 3D models of the prefusion virus protein. The new vaccine that used this prefusion version of the virus caused the body to produce antibodies 16 times as potent as the postfusion antibodies elicited by the old vaccine.
“I just wanted to know what the shape of the prefusion protein was—I wanted to know how it was folded and what it looked like,” Graham said. “The intent was just to understand it, but that led to a vaccine approach that looks like it's probably going to work.”
In fact, the discovery of the prefusion structure proved foundational to the work Graham’s lab subsequently began on coronaviruses like the severe acute respiratory syndrome and Middle East respiratory syndrome (MERS) viruses, both of which had previously caused worrisome outbreaks around the world. Some of this work was done in continued collaboration with McLellan, who had later joined the faculty at the Geisel School of Medicine at Dartmouth College (Hanover, New Hampshire) and now runs a lab at the University of Texas at Austin (Austin, Texas).
“After we had the RSV breakthrough, Dr. McLellan and I decided that coronaviruses were similar enough to RSV and there was no structural information for them,” Graham said. “That was a good area to work in because it was a wide-open field and it needed to be done.”
At that point, in 2014, Corbett joined the VPL as a senior research fellow and plunged full speed into understanding how the antibodies that bind to different forms of coronavirus spike proteins block infection. She also started developing a way for her team to quickly and reliably develop antigen proteins that could be tweaked to match each virus, as well as a method to deliver the instructions for making these proteins to cells via mRNA. The VRC was already gearing up for clinical trials with Moderna to test an mRNA vaccine against Nipah virus, which the lab had developed in parallel with an mRNA vaccine against the coronavirus that caused the 2012 MERS outbreak. As a result, by the time COVID-19 emerged, the VPL was poised to switch gears to a vaccine for COVID-19 and hit the ground running.
Inspiring Communities
In addition to her work on the Moderna COVID vaccine, Corbett has worked tirelessly on educating the public about vaccination and addressing vaccine hesitancy, particularly in communities of color. For the past year, her spare time has been filled with everything from television appearances and interviews to personally reassuring individuals and answering their questions. She even accompanied a young man and his mother to get their shots after the man expressed concerns about the COVID-19 vaccine at an MSNBC town hall. At the same time, Corbett hopes her newfound celebrity might motivate young people—especially women and minorities—to pursue careers in science.
“I try to tell my story because it’s not just that I don't even look like a scientist, but my background would suggest I could not be a scientist, ever,” Corbett said. “So I hope that people start to see that there's talent in different places.”

CREDIT: CHIA-CHI CHARLIE CHANG
President Joe Biden visits Barney Graham (foreground, left) and Kizzmekia Corbett (far right) at the VRC in February 2021.
“20 Years of Work for a Thousand People”
As the frenzied rush to develop a safe and effective COVID-19 vaccine winds down, both Graham and Corbett are planning their futures. Graham recently retired and hopes to devote his time to improving communication and education about science and technology here and around the world. In particular, he’d like to see the establishment of more research capacity in low- and middle-income countries and improved dissemination of information and technology.
“I’ve been stunned by the lack of information and understanding,” Graham said. “We need to improve community education.”
In June 2021, Corbett left NIH to lead her own laboratory at Harvard T.H. Chan School of Public Health (Boston), where she’ll continue to study coronaviruses and “do work on things that hopefully will surprise people,” she said.
She’ll have plenty of options. As Graham points out, there are 26 different families of viruses known to infect humans.
“You should pick something within those 26 families that you find really interesting,” Graham advised her. “It may end up being the cause of the next big pandemic, or it may not, but there’s at least 20 years of work for a thousand people.”
This story is adapted from one that originally appeared on the NIH “I Am Intramural” blog at https://irp.nih.gov/blog.
Podcast From the IRP
Dr. Kizzmekia Corbett—The Novel Coronavirus Vaccine

Melissa Glim, a science writer and health care communications professional, has written about topics from Alzheimer disease to women’s health, covering basic science to patient education to policy and advocacy. In her spare time, she loves making hats and jewelry, swing dancing, and writing the occasional children’s book, although most of the time, she’s waiting upon her beloved fox terrier, Tilly.
This page was last updated on Tuesday, February 1, 2022
