Restoring the Flow of Precious Saliva
IRP Researchers Aim to Regenerate Damaged Salivary Glands

IRP researchers are working on ways to restore natural saliva production in patients whose salivary glands have been damaged by autoimmune illnesses or radiation treatment for cancer.
It’s easy to take our saliva for granted. Most people have so much of it that they think nothing of spitting it out into a trash can when they finish chewing a stick of gum. Perhaps only people who have lived without it truly understand the great gift that is a perpetually moist mouth.
“Persistent dry mouth causes lots of problems with quality of life, and people forget how important saliva is until they lose it,” says IRP senior investigator Matthew Hoffman, B.D.S., Ph.D.
In honor of World Oral Health Day on March 20, a celebration of scientific efforts to reduce the burden of oral disease, I talked with Dr. Hoffman about his lab’s efforts to understand the biology of salivary gland dysfunction and translate that knowledge into treatments that bring relief to the many people suffering from it.
Genetic abnormalities, radiation therapy for cancer, and certain autoimmune diseases can cause the salivary glands to degrade or reduce their output, leading to chronic dry mouth, or ‘xerostomia.’ The condition is not only uncomfortable, but it can make tasting, chewing, and swallowing food difficult. It can also increase bacterial growth in the mouth, which leads to dental disease and infections.

Most patients with chronic dry mouth resort to sipping water constantly as a stopgap solution for their lack of saliva.
Treatments for dry mouth are currently limited and primarily focus on reducing symptoms and discomfort. Most people with damaged salivary glands end up carrying water to sip constantly. They also often use a saliva substitute, which is akin to a slightly sticky mouthwash that can coat the mouth and hold in moisture. In addition, there are a few medications available that rev up saliva production in the mouth’s salivary glands, but because these treatments affect glands throughout the body, they turn on all the faucets, leading to problems with excessive tears, sweating, and overheating.
“Radiation oncologists tell us they cure their patients’ cancers, but then their patients come back and say, ‘Hey, you’ve ruined my life,’” Dr. Hoffman says.
However, with the help of his IRP colleagues, Dr. Hoffman’s lab has made significant headway toward restoring function to damaged salivary glands. Their work focuses in particular on the damage caused by radiation therapy for head and neck cancers. By learning about how immature cells in the mouth interact with the nerves around them to grow into functioning salivary glands, Dr. Hoffman hopes to develop treatments that will repair and regrow that tissue.
“It’s like a developmental dance where you have one set of nerve cells growing and giving cues to the salivary gland cells, and the nerves growing and wrapping around the gland and providing signals for growth, development, and specialization,” explains Dr. Hoffman.
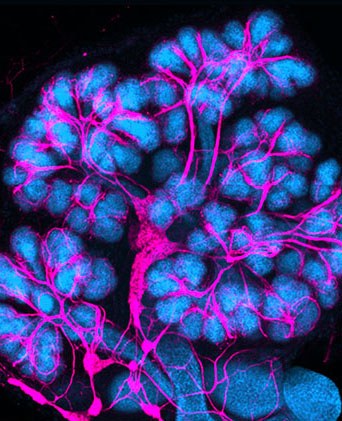
This image from Dr. Hoffman’s lab shows a developing mouse salivary gland with the nerves colored in pink. As development proceeds, neurturin helps guide the nerves to wrap around the salivary gland tissue.
Dr. Hoffman and his colleagues discovered that this process is guided by a particular protein called neurturin, which regulates the synthesis, growth, maturation, and survival of neurons. In experiments published in 2013, they showed that they could restore salivary gland function by adding neurturin to damaged salivary gland cells grown in the lab, as well as by injecting the protein into irradiated salivary glands in mice.1
Since then, Dr. Hoffman’s team has been exploring approaches to regenerating the salivary glands using both gene therapy and stem cells, immature cells that can morph into a variety of more specialized cell types. When Dr. Hoffman and his colleagues began their experiments, his IRP colleagues Bruce Baum, D.M.D., Ph.D., and John Chiorini, Ph.D., were running first-in-human gene therapy trials at the NIH Clinical Center with the aim of repairing salivary glands damaged by radiation, which can cause genes that are key to their function to stop working. Dr. Hoffman’s group decided to align their work with that project, using the same gene delivery system to insert an intact copy of the gene that produces neurturin into isolated salivary gland cells, with the ultimate aim of eventually testing their gene therapy in trials just like those being run by Dr. Baum and Dr. Chiorini.
“We were able to piggyback onto the expertise that was already here from the human gene therapy trials that were already ongoing,” says Dr. Hoffman.
The big discovery from that effort, Dr. Hoffman says, was that not only is it possible to restore salivary gland function using gene therapy, but the therapy can also be used preventively if it is initiated before radiation treatment.2,3
“In fact, it worked better to do it that way,” Dr. Hoffman says.

Dr. Matthew Hoffman
Dr. Hoffman’s team has also attempted to grow new salivary gland cells using stem cells. By manipulating stem cells taken from the salivary glands of mice and humans, the researchers found they could grow clusters of cells and coax them to start looking and behaving like salivary gland cells. 4,5 This work raises the tantalizing possibility of using a person’s own stem cells to regrow their damaged salivary glands.
While Dr. Hoffman’s ultimate goal is to create treatments that regenerate the salivary glands, his team’s research has also unlocked a much greater understanding of how to stimulate the growth and renewal of damaged tissue in general. As such, he hopes that his colleagues and the young scientists he mentors will be able to apply what they have learned in many different ways.
“As biologists, we can ask so many questions in our research, which opens up so many new avenues of thought and questioning that we could spend all our lives studying it,” says Dr. Hoffman. “But we've really been trying to push it toward the clinic. We’re not there yet, but we can certainly see a potential path forward.”
Subscribe to our weekly newsletter to stay up-to-date on the latest breakthroughs in the NIH Intramural Research Program.
References
[1] Parasympathetic stimulation improves epithelial organ regeneration. Knox SM, Lombaert IM, Haddox CL, Abrams SR, Cotrim A, Wilson AJ, Hoffman MP. Nat Commun. 2013; 4, 1494. doi:10.1038/ncomms2493.
[2] Neurturin gene therapy protects parasympathetic function to prevent irradiation-induced murine salivary gland hypofunction. Ferreira JN, Zheng C, Lombaert IMA, Goldsmith CM, Cotrim AP, Symonds JM, Patel VN and Hoffman MP. Mol Ther Methods Clin Dev. 2018; 9:172-180. doi: 10.1016/j.omtm.2018.02.008.
[3] CERE-120 Prevents Irradiation-Induced Hypofunction and Restores Immune Homeostasis in Porcine Salivary Glands. Lombaert IMA, Patel VN, Jones CE, Villier DC, Canada AE, Moore MR, Berenstein E, Zheng C, Goldsmith CM, Chorini JA, Martin D, Zourelias L, Trombetta MG, Edwards PC, Meyer K, Ando D, Passineau MJ, Hoffman MP. Mol Ther Methods Clin Dev. 2020; 18:839-855. doi: 10.1016/j.omtm.2020.07.016.
[4] Submandibular parasympathetic gangliogenesis requires Sprouty-dependent Wnt signals from epithelial progenitors. Knosp WM, Knox SM, Lombaert IMA, Haddox CL, Patel VN and Hoffman MP. Dev Cell 2015 Mar 23; 32(6):667-77. doi: 10.1016/j.devcel.2015.01.023.
[5] Neurturin-containing laminin matrices support innervated branching epithelium from adult epithelial salispheres. Vining KH, Lombaert IMA, Patel VN, Kibbey SE, Pradhan-Bhatt S, Witt RL, Hoffman MP. Biomaterials 2019 September; 216,119245. doi:10.1016/j.biomaterials.2019.119245.
Related Blog Posts
This page was last updated on Tuesday, May 23, 2023
