IRP’s Andre Nussenzweig Elected to National Academy of Medicine
NIH Researcher Recognized for Investigation into Genomic Stability

IRP senior investigator Andre Nussenzweig was elected to the National Academy of Medicine in 2019 in recognition of his research on a form of DNA damage that can both lead to cancer and be used to treat it.
The National Academy of Medicine (NAM), first established in 1970 by the National Academy of Sciences as the Institute of Medicine (IOM), is comprised of more than 2,000 elected members from around the world who provide scientific and policy guidance on important matters relating to human health. Election to the NAM is considered one of the highest honors in the fields of health and medicine and recognizes individuals who have not only made critical scientific discoveries but have also demonstrated a laudable commitment to public service.
IRP senior investigator Andre Nussenzweig, Ph.D., was one of four IRP researchers recently elected to the NAM. Dr. Nussenzweig leads the Laboratory of Genome Integrity at the National Cancer Institute (NCI), where he studies how cells repair a form of DNA damage called a double strand break (DSB). This type of insult, which severs both strands of the double-stranded DNA molecule, is one of the most dangerous. If not repaired properly, DSBs can kill cells or cause DNA to rearrange in ways that are associated with cancer. Moreover, while DSBs can be caused by chemotherapy drugs and radiation, they can also happen by random chance during the course of normal cellular processes. Intriguingly, not all parts of the DNA molecule are equally susceptible to this form of damage.
“It’s not always random,” Dr. Nussenzweig explains. “There are some parts of the genome that break more frequently than others, and we’ve been trying to understand the reasons why these regions are so fragile.”
When Dr. Nussenzweig received his Ph.D. from Yale University in 1989, few could have predicted that he would spend his career studying DNA and cancer. After all, his degree was in atomic physics, and his next career move was a postdoctoral fellowship in Paris under the mentorship of a French physicist who would go on to win the Nobel Prize in Physics. However, after finishing up in France, Dr. Nussenzweig began his pivot to biology by taking a position in the department of medical physics at Memorial Sloan-Kettering Cancer Center in New York City.
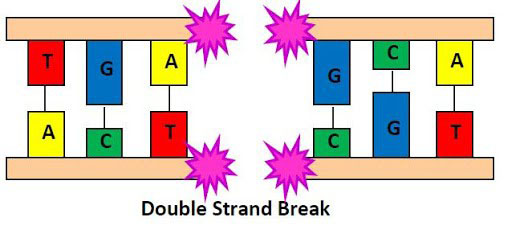
Illustration of a DNA double strand break (DSB). Image source: http://ozradonc.wikidot.com/types-of-dna-lesions
“That department combined physics and biology, so it was a great opportunity to transition into a new field,” he says. “They were basically doing radiation biology, so with my background, the pivot didn’t see so far-fetched, and they were sympathetic to my distinct perspective and desire to explore new research areas. I ended up working on a project related to DNA damage and repair, which seems fortuitous in retrospect.”
Once Dr. Nussenzweig began learning about DNA damage and repair, he was hooked. With the help of his IRP colleagues, he has produced important insights into what happens when these genetic injuries occur. This includes understanding how cells select between different ways to repair DNA and investigating how scientists and clinicians can exploit that ‘choice’ to prevent cancers from becoming resistant to chemotherapy, which kills cancer cells by breaking apart their DNA. His group has also developed a way to track the occurrence of DSBs, a technique that is now so in-demand among scientists that Dr. Nussenzwieg has a continuous stream of outside researchers visiting his lab to receive hands-on instruction on how to perform it.
Dr. Nussensweig’s lab created a method of pinpointing the precise locations of DSBs, a welcome development for researchers using CRISPR/Cas9 to edit DNA. Image credit: Ernesto del Aguila III, NHGRI
Part of this interest stems from the fact that Dr. Nussenzweig’s procedure is extremely useful for scientists leveraging another hot new technique: the gene editing platform known as CRISPR/Cas9, which allows scientists to modify DNA by breaking it apart at specific locations and subsequently inserting or removing DNA sequences at those sites. However, CRISPR/Cas9 sometimes also cuts DNA at locations other than those researchers want to target. Using the technique developed by Dr. Nussenzweig’s lab, scientists can determine exactly where CRISPR/Cas9 is severing DNA molecules to ensure it is doing so in the right places.
As an investigator at the NCI, Dr. Nussenzweig’s work also has important implications for the treatment of cancer. Different chemotherapies fight cancer by attacking their DNA at very specific locations, so identifying which parts of the genome are vulnerable to damage could help us better understand and improve chemotherapy treatment. In addition, the BRCA1 and BRCA2 genes associated with hereditary breast and ovarian cancer produce proteins that help repair broken DNA, making Dr. Nussenzweig’s research useful for understanding and treating cancers caused by mutations in those genes.
Looking back on his career so far, although he initially spent a great deal of time learning physics, Dr. Nussenzweig has no regrets about his leap to biology.
“What I find particularly exciting about biology is that you’re always finding new and unexpected phenomena,” he says. “However, some of these are hard to explain, and when it comes down to making predictions, you usually find that you’re wrong most of the time. This means that, unlike in physics, we still don’t have a good grasp of what’s going on in biology. That makes biology research very exciting and fast-moving, but also perilous.”
Subscribe to our weekly newsletter to stay up-to-date on the latest breakthroughs in the NIH Intramural Research Program. To learn about other IRP scientists recently elected to the National Academy of Medicine, check out our profile of Dr. Luigi Notarangelo.
Related Blog Posts
This page was last updated on Monday, January 29, 2024
