IRP Breast Cancer Researchers Answer “Redditor” Questions
Reddit “Ask Me Anything” Commemorates Breast Cancer Awareness Month

Dr. Stanley Lipkowitz (left) and Dr. Alexandra Zimmer (right) of the NCI Women’s Malignancies Branch answered questions from the Reddit community about breast cancer.
Breast cancer touches the lives of millions of Americans every year. In 2019 alone, researchers expect more than 300,000 American women to be diagnosed with breast cancer, along with more than 2,600 men. Roughly one out of every eight American women will develop invasive breast cancer at some point in her lifetime, making it the second most commonly diagnosed cancer in American women.
On October 9, in recognition of National Breast Cancer Awareness Month, the NIH Intramural Research Program (IRP) partnered with the National Cancer Institute (NCI) to host a Reddit “Ask Me Anything” (AMA) with two prominent researchers in the NCI's Women's Malignancies Branch: Stanley Lipkowitz, M.D., Ph.D., and Alexandra Zimmer, M.D. Between Dr. Lipkowitz’s extensive knowledge of the cellular and molecular pathways involved in breast cancer and Dr. Zimmer’s expertise in the development of clinical trials for breast cancer treatments, the pair were able to offer intriguing insights on topics ranging from recent advances in breast cancer treatment to genetic and environmental factors that influence risk for the disease. Read on for some of the most interesting exchanges that took place, or check out the full AMA on Reddit.
Question (Q): “There have been a number of advancements in the field of oncology and immunology over the past decade (e.g. immune checkpoint inhibitors, CAR-T cells, metabolic pathway modulators, monoclonals, etc). These new techniques and avenues have promised to improve patient outcomes and most importantly patient quality of life, but thus far they have been mostly used on final stage or as secondary treatments. Might we see chemotherapy replaced as the standard of care within the next decade?”
Alexandra Zimmer, M.D.: “A lot of new targeted therapies are under development. This is great because we can tackle different mechanisms of cancer development and have responses we were not able to see before. However, the idea for now has been to add options to therapy and build on what we already have available. Trials have been developed to add these new agents to some form of chemotherapy that we know is effective in cancer treatment already. A lot of these new targeted agents actually need the help of chemotherapy preparation to work better, like some protocols with CAR-T cells. So, I don’t see that we’ll get rid of chemotherapy in the near future, but we’ll learn to use it better in combination with therapies such as immunotherapy.”
Q: “What’s the deal with BRCA1 on a biochemical/genetic level? Senior undergrad bio major here."
Stanley Lipkowitz, M.D., Ph.D.: “I hope I am not doing your homework for you haha. BRCA1 is a gene that plays a role in repair of DNA damage. It was first identified in families with a high risk of breast cancer. People from these families who are at high risk of developing breast cancer have one normal copy and one mutant copy of the gene where the mutant copy is defective in DNA repair function. Preclinical studies have shown that losing both copies of the gene is not compatible with normal development and survival of the fetus, but having one normal copy of the gene is generally sufficient for normal DNA repair.
“There is a risk of any gene in a cell being mutated/inactivated, but losing both copies of the gene to mutation is an unlikely event. In people who already have one copy that is not functioning, the loss of the second copy leads to defects in DNA repair, causing mutations, and this is what leads to the development of cancer. It is much more likely to lose one copy of a gene than two, so this is why the risk of cancer is higher. For example, if a copy of a gene has a 1 in a million chance of becoming mutated/inactivated in any cell, then the chance that both would become mutated/inactivated in a cell is 1 in a million x 1 in a million, or 1 in a trillion. So if you start out with one abnormal copy, then the chance of a cell winding up with two mutant copies is a million times higher than in a cell that starts with two normal copies.
“There are many good articles and review articles on the function of BRCA1 at the molecular level and I would recommend starting there.”
Q: “I was wondering, the numbers for men are significantly lower than women getting breast cancer, do we know if this is purely because they have less breast tissue or is there another maybe hormonal component? Also to follow on, does breast size (for both men and women) have an impact on likelihood someone will get breast cancer? Thanks very much for your work!”
Dr. Lipkowitz: “For men, the incidence of breast cancer is about 100-fold lower than in women (i.e. about one percent of patients with breast cancer are men). I think there are several factors as you suggest. First, there is less breast tissue, but there is clearly a hormonal component as well. In women, hormones cause cycles of proliferation in breast cells throughout their reproductive lives, and this contributes to the risk of breast cancer whereas in men there is no such cycling in the breast.
“In terms of the relationship between breast size and breast cancer, there have been studies that are retrospective that suggest that patients with larger breasts do have an increased risk of breast cancer (and again that is part of the answer for men as indicated above). However, this needs to be studied in more detail in prospective studies and other factors which could affect breast size such as hormone levels or obesity also contribute to risk.”
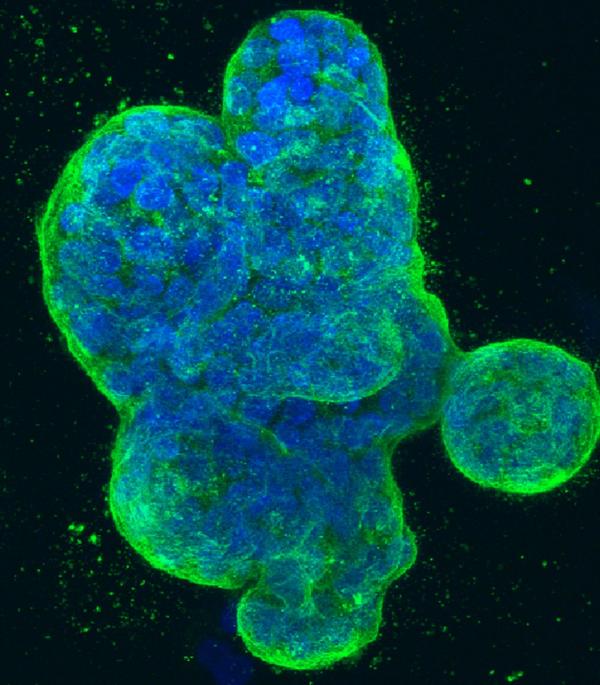
Three-dimensional culture of human breast cancer cells, with DNA stained blue and a protein in the cell surface membrane stained green.
Q: “Has breast cancer become more common as we have become more obese, and is there a causal link?”
Dr. Zimmer: “Obesity increases all sorts of health problems and mortality overall. It has been linked to increased risk of different types of cancers, including breast cancer, especially in postmenopausal women. The reason for that is likely the fact that most of the estrogen in postmenopausal women comes from the conversion of androgens to estrogen in fat tissue; having more fat tissue increases production of estrogen. There have also been studies suggesting that obesity increases inflammatory cells in the breast that may contribute to breast cancer risk or progression.”
Q: “Estrogen cause epigenetic changes in breast cancer. Do all types of oral contraceptives in premenopausal women and hormone replacement therapy in menopausal women cause epigenetic changes that influence the progression of breast cancer?”
Dr. Lipkowitz: “Estrogen drives expression of genes that contribute to the growth of normal breast cells and the growth of estrogen receptor-positive breast cancers. Oral contraceptives do increase the risk of developing breast cancer by 1.2- to 1.3-fold while taking the drugs and for a short time after that. By comparison, having a germline mutation in a gene such as BRCA1 or BRCA2increases risk by approximately 10-fold. In terms of hormone replacement therapy (HRT) in postmenopausal women, the risk of breast cancer is primarily associated with HRT that contains both estrogen and progestins. HRT with only estrogen is not associated with increased risk of breast cancer. Since growth of hormone receptor-positive breast cancers is driven at least in some part by estrogen, the use of estrogen-containing therapies should be avoided in patients who have breast cancer if it is estrogen receptor-positive.”
Q: “Do you all advocate for individual private genetic testing for genes like BRCA?”
Dr. Lipkowitz: “I would recommend that anyone being tested for inherited genetic abnormalities do this by seeing a genetic counselor to discuss the screening before it is done and to discuss the results once the test is completed.
"The concerns I have with private testing are that: 1) The tests are not necessarily comprehensive. For example, in some BRCA testing in the private genetic testing services they are testing for a small subset of mutations so that a negative test does not necessarily mean the gene is normal, only that you do not have one of the tested mutations. 2) It is not clear that the results are delivered to the patient by someone who is well-trained to discuss the meaning of the results and what the patient should do about this.”
Subscribe to our weekly newsletter to stay up-to-date on the latest breakthroughs in the NIH Intramural Research Program.
Related Blog Posts
This page was last updated on Monday, January 29, 2024
