Experimental Therapy Reverses Neurological Effects of Malaria
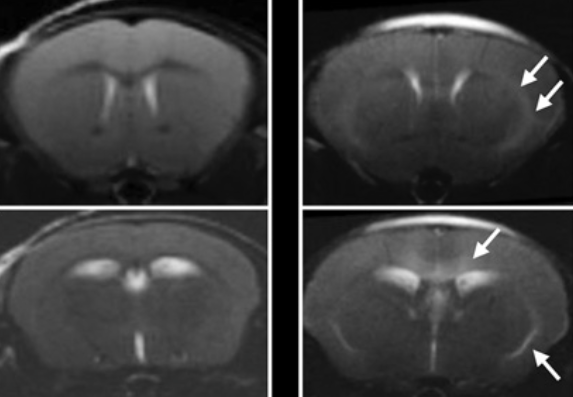
In a new IRP study, MRI scans showed abnormally bright areas in the brains of mouse models of cerebral malaria (right) compared to healthy mice (left), suggesting the illness had harmed the blood brain barrier. An experimental therapy co-developed by IRP scientists reversed this damage.
For Americans and others living outside the tropics, a mosquito bite is nothing more than an itchy inconvenience, but for billions of others, it can lead to a life-or-death battle with malaria. In some cases, the illness can wreak havoc on the brain. A new IRP study has used magnetic resonance imaging (MRI) technology to demonstrate that an investigational therapy can reverse that damage in mice.1
Over 500 million cases of malaria occur annually, and a small subset of these patients — mostly young children — develop cerebral malaria. In those individuals, the illness damages the blood-brain barrier, a structure that prevents various chemicals, toxins, and pathogens like bacteria from entering the brain. Between 15 and 25 percent of patients with cerebral malaria die despite treatment with antimalarial drugs2, and many survivors suffer from persisting neurological problems like blindness and epilepsy.
“The countries where cerebral malaria mainly occurs are generally medically underserved: the medical system is not well-developed with limited access to MRI scanners or intensive care units for treating patients,” says IRP investigator Dima Hammoud, M.D., the new study’s senior author. “Because we don’t understand very well what’s going on in the brains of those patients, we can’t really figure out the best treatment options.”
Recently, studies examining animal models of cerebral malaria have provided evidence that the disease is associated with high levels of a chemical called glutamate in the brain. High levels of glutamate can harm the blood-brain barrier. In their new study, Dr. Hammoud and her collaborators used MRI to document the brain changes that occurred in mouse models of cerebral malaria before and after they were given a drug designed to reduce glutamate levels in the brain. The therapy was developed by Johns Hopkins University School of Medicine researchers based on a compound that the lab of IRP senior investigator Susan Pierce, Ph.D., had shown to be effective in treating cerebral malaria in a mouse model.3
“Dr. Pierce’s group had shown that there was a benefit from the drug — the animals improved clinically — but nobody had been able to show the effects of the drugs visually,” Dr. Hammoud explains. “With imaging, you’re able to identify brain pathology very clearly when the animals are sick and how their disease is reversed after treatment.”
In collaboration with Dr. Pierce and her team, Dr. Hammoud’s research group first used MRI to examine the animals’ brains before treatment. A blood-brain barrier damaged by malaria allows fluid to leak out of blood vessels and into the brain, leading to potentially lethal brain swelling and other symptoms like seizures. The MRI scans showed that the disease increased the amount of fluid in the animals’ brains and provided evidence that this fluid came from leaking blood vessels. The researchers also injected a non-toxic chemical into the mice that is commonly used to enhance MRI images, and then used MRI to visualize bright areas in the animals’ brains caused by the chemical leaking out of blood vessels. As expected, MRI scans of healthy animals did not show any evidence of fluid accumulation in the brain or blood-brain barrier disruption.
While cerebral malaria is invariably lethal without treatment in mice, a majority of the animals given the experimental drug recovered from the illness. Post-treatment MRIs showed that the fluid buildup in the brain seen on earlier scans eventually disappeared in the mice that responded to the treatment. The fact that the drug reversed blood-brain barrier disruption even though it was given nearly a week post-infection is highly relevant, Dr. Hammoud says, because most patients with cerebral malaria don’t arrive at the hospital until the late stages of their disease.
“At this point, there isn’t really much available for those kids except supportive treatment: handling the seizures and giving them antimalarial drugs,” Dr. Hammoud says. “An additional drug like the one we are testing might have a lot of benefit for those vulnerable patients.”
Subscribe to our weekly newsletter to stay up-to-date on the latest breakthroughs in the NIH Intramural Research Program.
References:
[1] MRI demonstrates glutamine antagonist-mediated reversal of cerebral malaria pathology in mice. Riggle BA, Sinharay S, Schreiber-Stainthorp W, Munasinghe JP, Maric D, Prchalova E, Slusher BS, Powell JD, Miller LH, Pierce SK, Hammoud DA. Proc Natl Acad Sci U S A. 2018 Dec 4. pii: 201812909. doi: 10.1073/pnas.1812909115. [Epub ahead of print]
[2] Severe malaria. WHO. Trop Med Int Health. 2014. 19:7-131. doi:10.1111/tmi.12313.
[3] Targeting glutamine metabolism rescues mice from late-stage cerebral malaria. Gordon EB, Hart GT, Tran TM, Waisberg M, Akkaya M, Kim AS, Hamilton SE, Pena M, Yazew T, Qi CF, Lee CF, Lo YC, Miller LH, Powell JD, Pierce SK. Proc Natl Acad Sci U S A. 2015 Oct 20;112(42):13075-80. doi: 10.1073/pnas.1516544112. Epub 2015 Oct 5.
Related Blog Posts
This page was last updated on Wednesday, May 24, 2023
