A Country-Spanning Cancer Collaboration
Scientific Team-Up Identifies Source of Tumor Drug Resistance
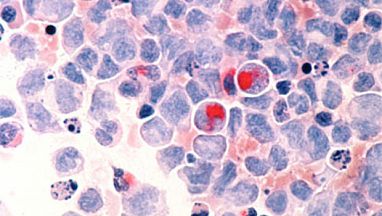
A recent collaboration between IRP researchers and scientists at Cincinnati Children’s Hospital Medical Center revealed how acute myeloid leukemia cells (pictured above) become resistant to certain treatments.
It’s an unfortunate reality that nearly everyone knows somebody whose life has been affected by cancer. However, a discovery by two researchers who met by chance years ago might one day help more cancer patients overcome their disease. Two scientific teams led by the IRP’s Craig Thomas, Ph.D., a group leader at the NIH’s National Center for Advancing Translational Sciences (NCATS), and Daniel Starczynowski, Ph.D., of Cincinnati Children’s Hospital Medical Center, recently published a study describing a possible breakthrough in the fight against acute myeloid leukemia (AML), a form of cancer responsible for nearly 11,000 deaths per year in the United States.
Nearly fifteen years ago, Dr. Thomas’ team began creating a collection of FDA-approved drugs and investigational compounds that included several molecules that inhibit a group of enzymes known as interleukin-1-receptor-associated kinases (IRAKs). The availability of these agents quickly spread from the NIH campus in Bethesda, Maryland, all the way to Cincinnati, Ohio, where Dr. Starczynowski caught wind of them through a mutual colleague.
“Our teams are in different cities, institutions, and have different scientific circles,” says Dr. Starczynowski. “It was a fortuitous collaboration which would not have happened without that initial interaction through a mutual colleague. It was that introduction which set us on this path.”
Dr. Starczynowski was studying a type of blood disorder called myelodysplastic syndrome (MDS), and his research suggested a role for IRAK inhibitors in MDS. Using the IRAK inhibitors collected by Dr. Thomas, Dr. Starczynowski was able to prove his theories were accurate and published his findings several years later. However, Dr. Starczynowski’s lab continued to study IRAK inhibitors and noticed that one of them demonstrated remarkable effects on AML cells. Intrigued by that data, Dr. Thomas and Dr. Starczynowski began to collaborate more closely and discovered that a subset of IRAK inhibitors had additional attributes that could prove useful against AML, including the ability to inhibit a protein called FLT3.

Dr. Craig Thomas
Like other cancer cells, AML cells begin as normal cells with healthy signaling pathways, but when a series of mutations causes a healthy cell to become cancerous, the newly cancerous cell co-opts those same pathways to divide rapidly and outcompete healthy cells. In some forms of AML, this process requires mutations in the FLT3 gene. These mutations send FLT3 into overdrive, producing chemical signals that direct AML cells to rapidly grow and divide. Targeted therapies exist for inhibiting this FLT3-based growth, but many cancers respond to these drugs by figuring out how to fuel their expansion in other ways.
Dr. Thomas’ and Dr. Starczynowski’s discovery of an IRAK inhibitor that also curbs the activity of FLT3 allowed them to examine if AML cells unable to use FLT3 to spur their growth were switching to an IRAK-dependent growth process. The IRAK signaling pathway is typically utilized by immune cells and had not previously been associated with the survival of AML cells, but the two scientists’ research teams were able to show that AML cells could use IRAK-based processes as a backup growth system.
“I would say that discovering this Achilles’ heel for this particular class of leukemia was in some ways unexpected,” says Dr. Starczynowski. “In fact, we still don’t fully understand how these cancer cells have the ability to hijack this IRAK-based pathway and survive in the absence of the main FLT3 signal they require to grow and reproduce.”
Dr. Thomas and Dr. Starcyznowski have high hopes that their dual IRAK-FLT3 inhibitor, or a version of it, has the potential to become a treatment for AML. Beyond that important goal, the discovery that IRAKs could be involved in cancer’s ability to resist therapies could also be used to develop more effective treatments for other forms of cancer that use an IRAK-dependent growth process.
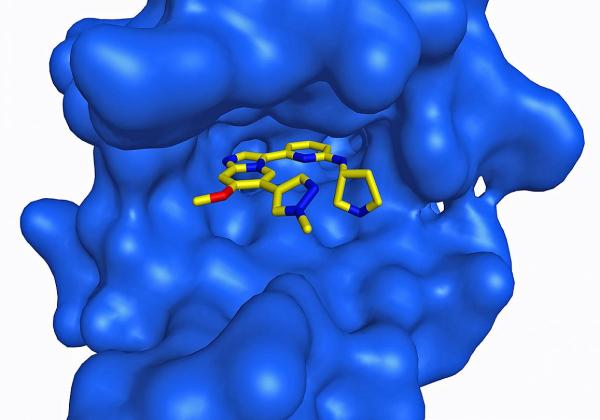
The chemical structure of a prospective drug sitting inside the protein kinase IRAK4. Image courtesy of Cincinnati Children’s Hospital Medical Center
This potentially pivotal study would not have been possible without the teamwork between the research groups led by Dr. Thomas and Dr. Starczynowski. As a whole, NCATS is known to be an integrated group without a disease-specific focus. Rather, NCATS studies the drug discovery process, which is an area of focus not traditionally part of academic research. As a result, the combination of Dr. Thomas’ drug development expertise and Dr. Starczynowski’s knowledge of cancer biology ultimately led to a discovery neither scientist could have made alone.
“The process of exchanging scientific ideas and brainstorming was really quite seamless between our two groups,” says Dr. Starczynowski. “We had open lines of communication and regularly were calling, emailing, and texting each other. I would jokingly say that I talk to Craig more often than some of my colleagues down the hall.”
Dr. Thomas agrees. “I have Dan on speed-dial,” he says.
Given the success of the partnership and the friendship that’s evolved from this collaboration, it is no surprise that Dr. Thomas’ and Dr. Starzynowski’s research teams will continue to work together. Currently, the two groups of scientists are continuing to study the FLT3- and IRAK-inhibiting compound they developed in an effort to translate their work into a new, more effective therapy for AML, offering hope to researchers, doctors, and cancer patients around the world.
“We’re excited,” says Dr. Thomas. “There is a lot of optimism around this research and the story doesn’t stop here.”
Subscribe to our weekly newsletter to stay up-to-date on the latest breakthroughs in the NIH Intramural Research Program.
Related Blog Posts
This page was last updated on Tuesday, May 23, 2023
