Three-Armed Antibody Could Offer Defense Against AIDS
Four Questions with Dr. John Mascola
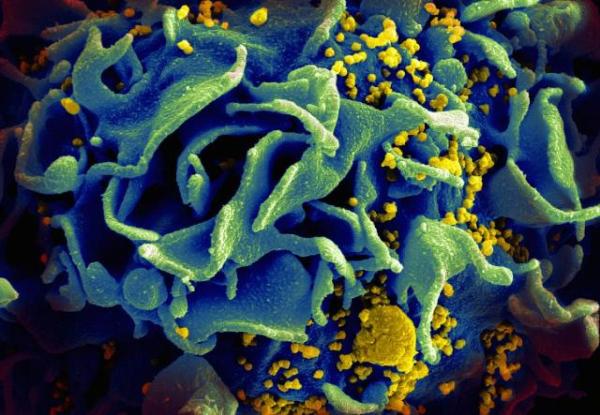
The HIV virus (yellow) weakens the immune system by attacking T cells like this one. Research led by the IRP's Dr. John Mascola could lead to a method to neutralize the virus and prevent this from occuring.
The disease known as human immunodeficiency virus, or HIV, attacks and destroys cells vital to the immune system. This leaves the millions of people living with HIV less able to fight other infections and can lead to an extremely severe form of immune system deficiency called acquired immunodeficiency syndrome (AIDS), which was responsible for nearly 770,000 deaths in 2018 alone. As of 2019, there are approximately 37.9 million people around the world living with HIV/AIDS.
Although HIV/AIDS has been recognized as a serious public health crisis, finding effective treatments, or a vaccine to prevent infection in the first place, is not a simple task. The HIV virus has many different types and strains — similar to the flu — which makes developing vaccines and treatments extremely challenging, as the virus is constantly changing. At the NIH, there are a number of ongoing collaborative research projects aimed at providing new options for those diagnosed with HIV/AIDS and those at risk for contracting the virus in the future.
A 2017 study led by IRP senior investigator John Mascola, M.D., sprung from a combined effort with a pharmaceutical company to build an antibody — the key element in the body’s adaptive immune system — that neutralizes HIV. While this may sound simple on paper, their approach in engineering the antibody was unique. The team developed an antibody with three arms, called a trispecific antibody, thereby offering three times the likelihood of neutralizing the virus compared to other antibodies, which typically contain fewer arms. As a result of their innovations, Dr. Mascola’s team found that its trispecific antibody successfully inhibited multiple strains of HIV and acted as a preventative treatment against an HIV-like virus in non-human primates, granting them complete immunity against the virus.
In honor of World AIDS Day this Sunday, December 1, I connected with Dr. Mascola to discuss his HIV research, explore what this study could mean for future treatments, and learn how he and his team are building on this research today.
What prompted you to conduct this research?
“For the past two decades, my focus as a scientist and a physician has been on HIV. The NIH’s Vaccine Research Center, which was established in 2000, has allowed me the opportunity to greatly expand on my HIV research because a major focus and priority of the entire institution is on developing a vaccine that could potentially prevent this disease. With this intention in mind, my collaborators and I set out to design that ‘three-in-one’ antibody, which has the potential to protect individuals from HIV by binding to three independent components of the HIV-1 virus’s outer coating.”

Dr. John Mascola
What was the most challenging aspect of this research?
“Designing the trispecific antibody molecule itself was particularly challenging. We were designing our antibody to have unusual features – three arms – versus natural antibodies that only have two arms. In order to design our three-armed antibody, we needed to find antibodies that were potent against HIV and compatible when we put them together in this trispecific form. It took us numerous tries and a lot of experiments to finally come up with the specific three-armed design that we needed.
“In general, HIV is a complicated infection, so there are naturally some challenges that researchers will face. Developing an effective vaccine for HIV has been a particularly difficult scientific obstacle because the virus tends to attack the immune system with various different strains. I like to tell people to think of HIV as diverse in the same way that the influenza virus is diverse. Each year, there are different forms of influenza, which is why people need to be vaccinated each year — because each year the vaccine changes to match the strains of the flu that are circulating around the world.
“In fact, HIV is actually much more diverse than influenza. If we want to use an antibody to prevent HIV infection, that antibody has to recognize all of the diverse variants of HIV that are circulating globally. Because our trispecific antibody has three arms, it has three times the potential to interact with genetically diverse types of HIV.”
What tools or collaborations were important in this research?
“Finding potent neutralizing antibodies was an important component of our study. Neutralizing antibodies function by binding to HIV and blocking its ability to infect a healthy cell. When these antibodies are circulating in a person’s blood, they could have the ability prevent the virus from actually infecting the individual at all. In our study, we had to find potently neutralizing antibodies by taking blood samples from HIV-infected donors.
“In order to isolate the neutralizing antibodies, we needed to screen volunteers infected with HIV infection to identify individuals who make unusually potent antibodies against the virus. The antibodies from these individuals were not only potent, but they were active against most of the different strains of HIV around the world. Thanks to these volunteers, we were able to make these antibodies in the laboratory and understand how they work against HIV.
“Once we were able to obtain and isolate antibodies from volunteers, we collaborated with the pharmaceutical company Sanofi, which had the technology to develop our trispecific antibody. Both the volunteers and the collaboration with Sanofi were critical to the success of our study.”
What impact has this research had on the field and how are you planning to build on it in the future?
"I’m excited to say that the trispecific antibody developed by our team has moved from studies in the laboratory and is now being studied in people – what we call phase 1 studies. Currently, the antibody is being examined in people diagnosed with HIV to make sure it is safe, and then to see if it can offer any therapeutic benefit against the disease.
“Future studies may test the concept of whether the trispecific antibody can actually prevent HIV infection and therefore provide benefits to healthy volunteers who are not infected with HIV but are at risk for HIV infection. There is an ongoing study with a single — not trispecific — antibody, and we will know more about the results within the next year. If those data are encouraging, then antibodies like this trispecific antibody could be used to prevent HIV infection, which is very exciting.
“We’re also continuing to work on developing better versions of this trispecific antibody — versions that are even more potent or could more broadly recognize the diverse strains of HIV.”
Head over to our Accomplishments page for more information on Dr. Mascola’s research, and subscribe to our weekly newsletter to stay up-to-date on the latest breakthroughs in the NIH Intramural Research Program.
Related Blog Posts
This page was last updated on Tuesday, May 23, 2023
