The SIG Beat
NEWS FROM AND ABOUT THE SCIENTIFIC INTEREST GROUPS
New SIG: Myeloid Malignancy Interest Group’s Inaugural Symposium

CREDIT: MARLEEN VAN DEN NEST
Scientists gathered on April 22, 2019, for the inaugural symposium of the new Myeloid Malignancy Scientific Interest Group.
The new Myeloid Malignancy Scientific Interest Group celebrated its launch with an inaugural symposium on April 22, 2019. Myeloid malignancies, such as myelodysplastic syndromes (MDS) and acute myeloid leukemia (AML), are rare, often highly fatal, cancers of the blood cells. The goal of this SIG is to intensify collaborations across the NIH communities and contribute to the ongoing growing momentum and interest in addressing these diseases and developing better treatments.
Myeloid malignancies are an area in which progress in cancer therapies has, until recently, been limited, and survival outcomes remain dismal. Recent advances in understanding the genomics of these diseases, as well as tumor immunology, are opening unpreceded opportunities for more-rapid translation of new therapies into the clinic. The prominent unmet needs in this field are MDS and secondary AML. These diseases are particularly suited for being studied in the NIH intramural research program due to their inherited and acquired genetic defects, relative rarity, and frequently subacute disease trajectory.
Readily available bone marrow samplings in these patients represents an ideal model for longitudinal study of cancer progression from initial genetic defects to a fully expressed clinical malignancy. Approximately 20,000 new cases of AML are diagnosed each year in the United States; of these, about 10 percent are related to prior cancer therapy and an additional 20 percent emerge from antecedent MDS. These two subsets of AML have the poorest prognosis and are not curable by the existing therapies, except in patients who successfully undergo allogeneic hematopoietic stem-cell transplantation (HSCT). Moreover, the incidence of MDS and secondary AML exponentially and dramatically rises in age groups above 65, in whom intensive therapies are poorly tolerated and HSCT is usually not an option.
There is also a small but increasingly recognized group of MDSs and AMLs that arise from inherited genetic diseases and evolve, often in young adulthood, into very aggressive leukemia for which pathogenesis and natural histories are poorly understood. All these entities have been studied in several research laboratories by investigators at the intramural NIH.
Symposium
Christopher Hourigan (NHLBI), one of the founders of the new SIG, welcomed everyone and reported that there were already 73 people on the SIG’s LISTSERV and that at least four institutes were represented as well as the FDA and extramural organizations. Deputy Director for Intramural Research Michael Gottesman explained the importance of having a program in myeloid malignancies—it’s complicated; there are interesting animal and genetic models; scientists are getting closer to understanding the origins of the myeloid diseases; and there are multiple modalities for treatments. Gottesman, who’s also the director of NCI-CCR’s Laboratory of Cell Biology, spoke about his research on the mechanisms of multidrug resistance in cancer. His lab identified the first ATP-binding cassette transporter and showed how it conferred multidrug resistance in cancer. The lab is trying to figure out the role of the transporters in AML.
Steven Pavletic, a senior clinician in NCI-CCR and also one of the SIG’s founders, gave an overview of myeloid-malignancies research at NIH. He explained that the SIG aims to bring together NIH MDS and AML researchers and to intensify collaborations among them to cure, treat, and prevent these diseases. He pointed out that there was a session on myeloid malignancies at the 2018 Research Festival.
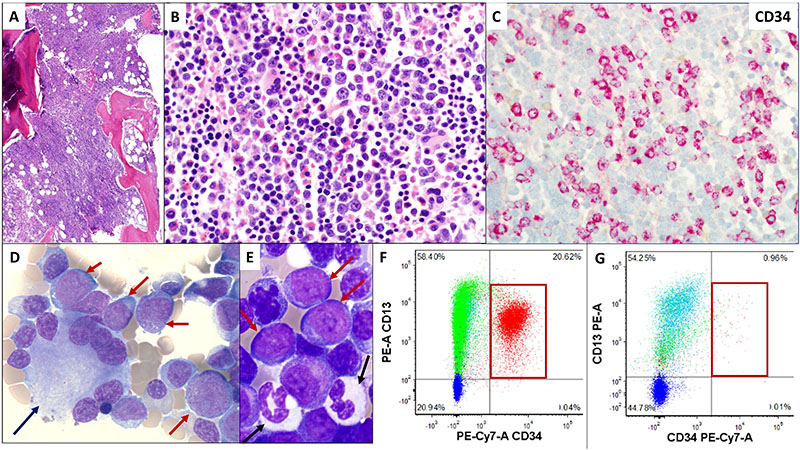
CREDIT: KATHERINE CALVO, CC
Bone marrow from an 81-year-old male with history of myelodysplastic syndrome who progressed to acute myeloid leukemia (AML). A) An excess of cells in bone marrow; B) an increased number of immature leukocytes; C) immunohistochemistry for myeloblasts expressing CD34 confirms acute leukemia; D) bone-marrow aspirate showing abnormal megakaryocyte (bone-marrow cell responsible for the production of platelets) with peripheralized, separated nuclear lobes (black arrow) and increased immature cells called myeloblasts (red arrows); E) marrow aspirate with dysplastic hypogranular neutrophils (black arrows) and increased myeloblasts (red arrows); F) flow cytometric analysis of marrow aspirate showing over 20 percent myeloblasts (red box); having at least 20 percent myeloblasts is required for a diagnosis of AML; (G) six weeks post treatment, on NIH protocol 17-H-0026 with a novel combination of pembrolizumab and decitabine, flow cytometric analysis of marrow shows less than one percent myeloblasts (red box).
Several researchers presented their work. Daniel Larson, a senior investigator in NCI-CCR, gave a talk titled “Myelodysplastic Syndromes—From Lab to Clinic” in which he described some of his research. Naoko Takebe (NCI) discussed the rationale for a phase 1 clinical trial involving a combination treatment with clofarabine and bortezomib for patients with solid tumors, lymphomas, and MDS. Case studies were presented by Takebe, Clinical Center investigators Alina Dulau-Florea and Katherine Calvo, and NHLBI investigator Hourigan. One of the cases highlighted was that of an 81-year-old man who had refractory MDS that evolved into AML. The patient participated in a combination drug trial that administered pembrolizumab and decitabine and he showed marked improvement in six weeks (a reduction in myeloblasts, from 20 percent to less than one percent).
For information about this SIG and instructions on how to join its LISTSERV newsletter, go to https://oir.nih.gov/sigs/myeloid-malignancy-interest-group. For other questions, contact Steven Pavletic (pavletis@mail.nih.gov).
For a complete list of SIGs, go to https://oir.nih.gov/sigs/view-name. To start a new SIG, contact OIRinfo@mail.nih.gov.
This page was last updated on Monday, April 4, 2022
