Sleep, Perchance to Research
NIHers Are Studying Sleep, Fatigue, and Circadian Rhythms
A sleep-deprived person may still function, but not as efficiently as someone who gets enough good-quality sleep, and they may be at increased risk for heart disease, kidney disease, diabetes, obesity, high-blood pressure, stroke, and a host of other problems. Lack of sleep may even affect one’s ability to learn and remember information.
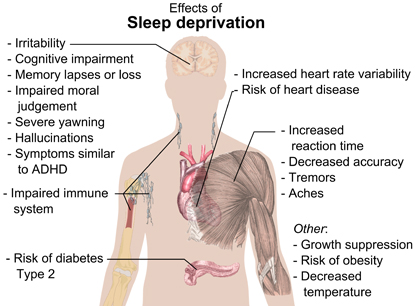
MIKAEL HÄGGSTRÖM, WIKIMEDIA COMMONS
Effects of sleep deprivation. Sleep plays an important role in physical health. Sleep promotes the healing and repair of heart and blood vessels, helps maintain a healthy balance of hormones, and plays a role in learning. Ongoing sleep deprivation is linked to increased risk of heart disease, kidney disease, diabetes, obesity, high blood pressure, stroke, and other problems.
“Elucidating the nuts and bolts of what goes wrong [in sleep] is the cutting edge for much of the [sleep-related] research that is going on” at NIH and elsewhere, said Michael Twery, director of NIH’s National Center on Sleep Disorders Research, which oversees the support of research and research training related to sleep disorders and stewards several forums that facilitate the coordination of sleep research across NIH, other federal agencies, and outside organizations.
At NIH, there are more than 50 researchers studying sleep, fatigue, and circadian rhythms. The NIH Catalyst interviewed four of them and provided descriptions of the work of many of the others.
The researchers themselves, when they reviewed the lists for accuracy, were surprised at how many intramural researchers were involved in sleep-related research and remarked that knowing who the others are might pave the wave for future collaborations.
The Mind’s Clock: David C. Klein
For someone who says, “I was never really interested in sleep” research, neuroendocrinologist David Klein (National Institute of Child Health and Human Development) has significantly contributed to the field by identifying the molecules and brain regions that regulate the internal clock in all vertebrates.
“I was interested in endocrinology, and the pineal gland had the shortest chapter [in textbooks] … so I figured I could make the biggest contribution,” he joked. The pineal gland is a small melatonin-producing structure in the center of the vertebrate brain. Melatonin, discovered by a team of researchers led by Yale dermatologist Aaron B. Lerner in 1958, is a hormone that regulates circadian rhythms.
When Klein joined the NIH in 1969, NIH neuroscientist Julius Axelrod was already investigating the synthesis of melatonin. “I was competing with a man with a Nobel prize,” said Klein. (Axelrod, who worked in the National Heart Institute and the National Institute of Mental Health, shared the Nobel Prize in Physiology or Medicine in 1970 for his discovery of the actions of neurotransmitters in regulating the metabolism of the nervous system.)
Soon, however, Klein made the breakthrough discovery that the daily rhythm of melatonin production is regulated by arylalkylamine N-acetyltransferase, an enzyme responsible for serotonin acetylation. Basically, this enzyme, which Klein coined the “timezyme,” controls melatonin production, turning it on and off very rapidly.
Klein playfully handled a large, brightly colored crystalline model of the “timezyme” while he explained its unique structure. Concentrations of “timezyme,” and subsequently melatonin, increase at night in all vertebrates. Because not all animals sleep at night, melatonin is not a simply a signal to sleep but truly a signal of time, even used for seasonal timing in some species.
NIGMS
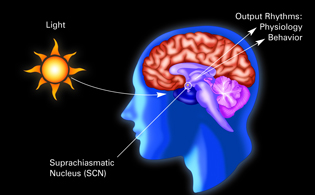
NICHD investigator David Klein and a colleague found that a tiny subunit of the hypothalamus called the suprachiasmatic nucleus (SCN) was essentially a circadian pacemaker, or what he calls “the mind’s clock.” The SCN helps control sleep by coordinating the actions of billions of miniature “clocks” throughout the body. These aren’t actually clocks, but rather are ensembles of genes inside clusters of cells that switch on and off in a regular, 24-hour cycle.
In collaboration with neuroanatomist Robert Moore, Klein found that a tiny subunit of the hypothalamus called the suprachiasmatic nucleus (SCN) was essentially a circadian pacemaker, or what Klein calls “the mind’s clock.” This brain region contains melatonin receptors and works as “the master oscillator” that keeps the circadian clocks in the body synchronized with one another and to the 24-hour day. The SCN also controls the endogenous sleep rhythms of when to sleep and for how long. If the SCN is destroyed, circadian rhythmicity is abolished as well as the ability to synchronize patterns of daily activity with the light cycle.
Klein believes he has influenced the field of sleep research by raising awareness of the SCN; in 1991, he, Moore, and a colleague co-edited Suprachiasmatic Nucleus: The Mind’s Clock, a book devoted to explaining the significance of the SCN.
Today, melatonin is a widely used, self-administered sleep aid. There are claims that melatonin helps you to fall asleep, stay asleep, and maintain healthy sleep patterns. However, Klein points out that many of these claims have not been scientifically proven and effects may not be similar in everyone.
Currently, Klein’s laboratory is focused on characterizing the transcriptome (the very small percentage of the genome that is transcribed into RNA molecules) of the pineal gland. Using high-throughput DNA and RNA sequencing techniques, they have found hundreds of genes that are significantly altered over a 24-hour cycle. These genes, some of which exhibit a 100-fold difference in day-night expression, control many functions including the fate and phenotype of pinealocytes, the cells responsible for producing melatonin.
The Link Between Obesity and Sleep: Giovanni Cizza
It seems that everything we do or eat these days is somehow linked to obesity. Sleep–both when we sleep and for how long—has been attributed to obesity in epidemiological studies and in controlled laboratory studies. For example, epidemiological evidence shows that shift workers weigh more than non–shift workers even after extenuating variables are taken into account. In a controlled laboratory setting, when healthy, lean male subjects are sleep deprived for four hours per night for five days, they develop insulin resistance with a 20 percent increase in ghrelin (a hormone that stimulate appetite) and a 20 percent decrease in leptin (a hormone that suppress appetite). However, little is known about how sleep disturbance causes obesity.

BILL BRANSON
NICHD investigator Giovanni Cizza (now at the FDA) spent a large part of his career addressing important questions on the relationship between sleep and obesity. Pictured: Cizza is standing next to a recruiting poster—for a sleep and weight study—that features Pablo Picasso’s painting of a woman sleeping in a chair.
Investigator Giovanni Cizza (National Institute of Child Health and Human Development) spent a large part of his career as a clinical investigator at the NIH addressing two important questions surrounding sleep and obesity. First, what happens to the metabolism of people who are sleep deprived for social reasons when they are given an opportunity to sleep longer? Second, why are individuals with narcolepsy (who cannot regulate their sleep cycle and so sleep at random times throughout the day) about 15 pounds heavier than healthy control subjects?
To answer the first question—How do sleep-deprived people respond to adequate sleep?—Cizza enrolled obese people who self-reported sleeping fewer than 6.5 hours per night in a study and coached them to increase their sleep time to at least 7.5 hours per night for 15 months. Before and immediately after the intervention, they spent time at the NIH undergoing many baseline and follow-up tests assessing metabolism, body weight, insulin sensitivity, hormone concentrations, and neurological function. Cizza found that sleeping longer improved the participants’ neurocognitive functions (such as memory) and executive functions (learning and decision making) by up to 10 percent. Some of the results on body weight and metabolism were published in the August 2014 issue of the electronic journal PLOS ONE. (PLOS ONE 9:e104176, 2014)
For the the second question—Why do individuals with narcolepsy weigh more than healthy control subjects?—Cizza hypothesized that individuals with narcolepsy have decreased energy expenditure compared with healthy control subjects. After all, mice with narcolepsy weigh more than healthy mice because they expend less energy and therefore burn fewer calories. The extra calories are stored as fat. To test the hypothesis in humans, Cizza has so far recruited about 20 subjects with matched control subjects and put them in a room-sized metabolic chamber for 24 hours to measure their oxygen consumption and carbon-dioxide production, which reflect energy expenditure. He’ll report his findings when the study is complete.
Cozying Up with Sleeping Flies: Susan Harbison
Susan Harbison didn’t foresee the day she would be meticulously measuring the genetics of sleep in flies when she started her career as an aerospace engineer analyzing structural stress factors on Navy helicopters. Later, after going back to school to get a Ph.D. in genetics and doing postdoctoral work in neuroscience and genetics, she found her calling—quantitative genetics.
Now she is an Earl Stadtman Investigator in the National Heart, Lung, and Blood Institute’s (NHLBI’s) Laboratory of Systems Genetics, where she is trying to derive computational models describing how gene networks influence sleep.

NHLBI
The recent recipient of a Presidential Early Career Award for Scientists and Engineers, NHLBI investigator Susan Harbison was recognized for her work into the genetic and environmental changes—such as drug exposure—affect sleep patterns in Drosophila (fruit flies). Since sleep in Drosophila has all the behavioral characteristics of mammalian sleep, she hopes that the identification of gene networks may have implications for humans.
She focuses on the Drosophila (fruit fly) model because so many powerful genetic tools exist to study it. Furthermore, sleep in Drosophila has all the behavioral characteristics of mammalian sleep. Immobile periods of five minutes or more and a drooping posture (resulting from muscle relaxation) define fruit-fly sleep. A fruit fly will try to make up for lost sleep. An increased arousal threshold is also observed—for instance, experimental vials need to be tapped with greater force to rouse a sleeping fruit fly, just as you might need to be forcefully shaken awake from a deep slumber. A fruit fly’s sleep cycle is diurnal, and fruit flies also spend a significant portion of their lives asleep just as we do, in some cases as much as a combined 15 hours in a 24-hour period.
“I measured things [such as] sleep duration, the number of sleep-bouts or naps, the average sleep-bout length, [and] waking activity, which is a measure of how hyperactive the flies are,” Harbison told NHLBI Director Gary Gibbons in a recent interview that appears on the NHLBI Web site.
To measure sleep in fruit flies, she used an infrared-based Drosophila activity-monitoring system. Each fruit fly is placed in a three-inch-long glass tube. “When the fly walks back and forth, he breaks the infrared beam, and that tells us whether or not he’s active,” Harbison explained to Gibbons. The data generate a series of text files that include numbers of counts per minute. “We can decipher [sleep phenotypes] from that.”
Harbison has generated some exciting results using a genome-wide association study (GWAS) in which she probed 2.5-million genetic variants in a collection of inbred fruit flies whose ancestors were captured in the wild. She identified single-nucleotide polymorphisms, many of which have human homologues that may be associated with natural variations in sleep.
Now the big problem facing Harbison is determining which candidate genes contribute most to sleep behavior. In fruit flies of identical genotypes, she found that sleep patterns were affected by changes in the environment. She also observed differences in sleep patterns between male and female fruit flies: Males have bursts of activity at dawn and at dusk that might be related to courtship behavior; females are active at a lower level throughout the day and take shorter naps than males do. Sleep deprivation also affects glycogen content in males and triglycerides in females.
Human sleep disorders are correlated with learning and memory impairment, neurological diseases, cardiovascular problems, and hypertension, to name a just a few. Within this complex web the question of whether sleep is needed for one particular function before all others remains a puzzle. “There’s not one theory of sleep that everyone is jumping on,” said Harbison. Indeed, the GWAS candidate genes identified in her work represent aspects of all the current theories on the need for sleep, providing no shortage of big questions to ask.
To listen to Harbison’s interview with NHLBI Director Gary Gibbons, go to http://1.usa.gov/1qjfX22. To view Harbison’s presentation that she gave on April 1, 2014, as part of the Demystifying Medicine series, go to http://videocast.nih.gov/launch.asp?18362.
Why Sleep? Carolyn Beebe Smith
Why do we need to sleep? Senior Investigator Carolyn Beebe Smith in the National Institute of Mental Health (NIMH) is exploring this essential question by imaging the brain during wakefulness and sleep and by correlating brain-protein metabolism with learning and memory.

It’s thought that sleep is needed to maintain, repair, and reorganize brain cells. In animals, the formation of brain proteins increases during sleep. Sleep also seems to enable synaptic remodeling processes that promote neuronal plasticity during development, learning, and memory formation.
Smith is conducting a clinical trial, using positron-emission tomography (PET), to examine the formation of brain proteins while people are awake, deprived of sleep, and asleep; and to assess brain-protein syntheses in waking and sleep combined with a learning task—a computerized visual-discrimination task. Participants are injected with a radiolabeled amino acid that is detectable by a PET scan. Persistence of radiolabeled amino acids in the brain indicates that they are being incorporated into new proteins. New protein synthesis serves as a correlate for the synaptic remodeling events required for learning and memory consolidation.
Some participants are allowed to nap after training and some are not. All are trained in the morning on the computerized visual-discrimination task and then tested eight hours later. Subjects who napped showed more improvement on the test than those kept awake. Moreover, preliminary results of PET scans performed during the nap indicate that protein synthesis is increased in a part of the primary visual cortex involved in the training. Smith's preliminary findings demonstrate that protein synthesis increases during memory consolidation and suggest that synaptic remodeling and neuronal plasticity may be key functions of sleep.
To view the presentation Smith gave on April 1, 2014, as part of the Demystifying Medicine series, go to http://videocast.nih.gov/launch.asp?18362.
A FEW OTHER NIHERS DOING RESEARCH ON SLEEP, FATIGUE, AND CIRCADIAN RHYTHMS
CLINICAL CENTER
Gwenyth R. Wallen, R.N., Ph.D. (chief, Nursing Research and Translational Science Section, NIH Clinical Center), is collaborating with NHLBI hematologists James Taylor VI and Caterina Minniti on a study to determine the prevalence of sleep disturbance and whether it’s associated with pain and depression in sickle-cell disease. They found that in 328 sickle-cell patients, more than 70 percent of adults had sleep disturbance; 21 percent showed evidence of clinical depression; and sleep disturbance and depression were correlated (BMC Psychiatry 14:207, 2014). In another clinical study, she is collaborating with NIAAA investigators (Vijay A. Ramchandani, David T. George, and Melanie Schwandt) looking to measure the prevalence of sleep disturbance in people with alcoholism undergoing inpatient alcohol detoxification. So far they have found more than a 90 percent prevalence of sleep disturbance (Fam Community Health 37:288-297, 2014). Their newest clinical study examines sleep and potential relapse across the continuum from inpatient alcohol treatment to post discharge into the community.
Leighton Chan, M.D. (senior investigator, Rehabilitation Medicine Department, NIH Clinical Center), is doing a natural history study of traumatic brain injury in which patients are asked about sleep-related issues. One questionnaire is designed to differentiate fatigue from clinical depression; the other measures daytime sleepiness. The data are being analyzed.
Lynn Gerber, M.D. (special volunteer and former chief, Rehabilitation Medicine Department, NIH Clinical Center; now a professor at George Mason University in Fairfax, Virginia, and director of research in the Department of Medicine at Inova Fairfax Hospital in Falls Church, Virginia), is collaborating with Leighton Chan and other NIH researchers to study the causes of functional loss and disability in chronic illness as well as the mechanisms and treatment of fatigue. In particular, she is interested in the associations between pain and function and fatigue in people with myofascial pain and in breast-cancer-related fatigue.
NATIONAL CANCER INSTITUTE
Mirit I. Aladjem, Ph.D., and Kurt Kohn, M.D., Ph.D. (senior investigators, Developmental Therapeutics Branch, NCI-CCR), created an extended computational model of a mammalian circadian clock to provide insight into the regulation of circadian rhythms and their potential role in cancer biology. The study was conducted in the context of describing the biochemical activities of the multifunctional protein sirtuin 1, which is involved in circadian rhythm regulation as well as in many other metabolic functions. Results from this model may also add to knowledge on the role of circadian rhythms on the toxicity and activity of therapeutics, including common cancer drugs.
Gordon Hager, Ph.D. (senior investigator and chief, Laboratory of Receptor Biology and Gene Expression, NCI-CCR), is doing work related to ultradian (a recurrent period throughout a 24-hour day) and circadian cycling of hormones and glucocorticoid receptors. He is also hoping to collaborate with Lynette Nieman (NICHD) to test the effects of pulsatile treatment (treatment delivered in pulses instead of continuous infusion) in Addison disease patients. Addison disease is a rare, chronic disorder in which the adrenal glands produce insufficient glucocorticoids and other hormones.
NATIONAL HUMAN GENOME RESEARCH INSTITUTE
Ann C.M. Smith, M.A., Honorary D.Sc. (adjunct principal investigator, NHGRI), is conducting a clinical trial to examine the effect of bright light or melatonin treatment on circadian sleep disturbance in children with a developmental disorder called Smith-Magenis syndrome (SMS). People with SMS have a disrupted sleep cycle that involves early waking, frequent daytime napping, and frequent nighttime awakenings. Melatonin is normally produced at night in healthy people, but people with SMS produce high concentrations of the hormone during the daytime and very low concentrations at night. This anomaly may affect behavior, mood, attention span, and sleep patterns. SMS is named for Smith—and Ellen Magenis—who, in 1982, identified the first two families with it.
NATIONAL HEART, LUNG, AND BLOOD INSTITUTE
Susan Harbison, Ph.D. (Stadtman Investigator, Laboratory of Systems Genetics, NHLBI). See interview.
Amisha V. Barochia, M.D. (staff clinician), Nargues Weir, M.D. (staff clinician), and Stewart Levine, M.D. (senior investigator, Laboratory of Asthma and Lung Inflammation, NHLBI), are conducting basic and clinical research on asthma, including a longitudinal observational study of severe asthma, which involves an overnight sleep study.
John Tisdale, M.D. (senior investigator, Molecular and Clinical Hematology Branch, NHLBI), Courtney Fitzhugh, M.D. (assistant clinical investigator, Laboratory of Sickle Mortality Prevention, NHLBI), and James Taylor are working on multiple strategies both in the laboratory and in the clinic to cure sickle-cell disease (SCD). Fitzhugh did a clinical study to determine whether the neuropeptide galanin is altered in subjects with SCD who have sleep disturbances. Taylor worked on a project with Gwenyth Wallen (CC) in which they found a high prevalence of sleep disturbance in patients with SCD.
James Taylor VI, M.D. (assistant clinical investigator, Genomic Medicine Section, NHLBI), in addition to the studies mentioned above, has been working on genome-wide genetic-association studies. Genetic as well as environmental factors play important roles in sleep disturbance. Taylor’s preliminary findings suggest that, in people with sickle-cell disease, there may be a genetic association on chromosome 13.
NATIONAL INSTITUTE ON ALCOHOL ABUSE AND ALCOHOLISM
Nora Volkow, M.D. (senior investigator, Laboratory of Neuroimaging, NIAAA; Director, NIDA), used positron-emission tomography to show that sleep deprivation reduced dopamine (DA) receptor availability (J Neurosci 32:6711–6717, 2012). DA is a key factor in the addiction process. There is increasing evidence that DA exerts a wake-promoting action and that DA receptors are involved in wakefulness. In addition, patients with Parkinson disease, who suffer from DA depletion, experience excessive daytime sleepiness. In future clinical studies, Volkow plans to assess whether there is a disruption of circadian DA rhythms in cocaine addiction and whether DA modulates the sensitivity of the brain’s reward network. She also plans to measure the glymphatic function (the waste-clearance system in the mammalian central nervous system) in the human brain during sleep and wakefulness and possibly investigate the effects of alcoholism, which disrupts sleep patterns.
Lorenzo Leggio, M.D., Ph.D. (clinical investigator and section chief, NIAAA and NIDA), Melanie Schwandt, Ph.D. (staff scientist), Vijay A. Ramchandani, Ph.D. (section chief), and Nancy Diazgranados, M.D. (staff clinician), are collaborating with Gwenyth Wallen, R.N., Ph.D. (CC), on a study of sleep disturbances in people with alcoholism who are undergoing inpatient alcohol detoxification. The study includes an assessment of whether sleep quality predicts relapse.
Matthew Pava, Ph.D. (postdoctoral fellow), and David Lovinger, Ph.D. (senior investigator and section chief), Laboratory for Integrative Neuroscience, NIAAA, are measuring brain and muscle electrical activity in mice to characterize how the endocannabinoid system modulates sleep and wake states. Endocannabinoid signaling is known to be disrupted by chronic exposure to alcohol or cannabinoid drugs. The researchers are trying to determine whether the signaling alterations underlie some aspects of sleep disturbances in alcohol- or cannabis-dependence disorders. Pava is also collaborating with David Kupferschmidt, Ph.D. (postdoc), to assess sleep in mice that lack the presynaptic protein Rim1a (Rab3-interacting in mitochondria 1-alpha).
NATIONAL INSTITUTE OF CHILD HEALTH AND HUMAN DEVELOPMENT
Giovanni Cizza, M.D., Ph.D. (special volunteer, NICHD; FDA; former investigator in NIDDK). See interview.
David Klein, Ph.D. (senior investigator and chief, Section on Neuroendocrinology, NICHD). See interview.
Margaret F. Keil, Ph.D, C.R.N.P. (director, Pediatric Endocrine Clinical Services, NICHD), is conducting a biobehavioral study that includes studying sleep patterns of children who were recently adopted from orphanages in other countries. She hopes to learn more about the effects of deprivation on neuroendocrine function, physical growth, and developmental outcomes, including cognitive and behavioral measures. In addition, she will examine nutritional intake and sleep patterns to determine their effect on growth and developmental outcome. In the future, she plans to assess the effect of cortisol dysregulation on sleep.
Lynnette K. Nieman, M.D. (senior investigator, Section on Reproductive Endocrinology, NICHD), is conducting a clinical trial on jet lag to determine whether taking cortisol, melatonin, or both can help alleviate jet lag and re-establish the body’s day-night cycle.
Jack A. Yanovski, M.D. (senior investigator, Section on Growth and Obesity, NICHD), is conducting a clinical trial to learn about the role of the PAX6 gene in sleep patterns in people with WAGR syndrome (a rare disease in which children develop a tumor of the kidney) or people with isolate aniridia (absence of the colored part of the iris). PAX6 may play an important role in the development of the pineal gland, the gland in the brain that produces melatonin, a hormone involved in sleep.
Paul Albert, Ph.D. (senior investigator and chief, Biostatistics and Bioinformatics Branch, NICHD), developed a statistical model to measure the unobserved sleep-wake cycle in a multiyear longitudinal study of adolescents. His methods are helping researchers measure sleep patterns and determine how they relate to future poor behavioral outcomes.
NATIONAL INSTITUTE OF DIABETES AND DIGESTIVE AND KIDNEY DISEASE
Yaron Rotman, M.D. (clinical investigator, Liver Diseases Branch, NIDDK), is conducting a clinical trial on the physiology of fatigue in patients with chronic liver disease. Fatigue is a common and often disabling symptom in these people, and sleep disturbance may play a role. His study looks at the body’s circadian rhythms to see whether problems with them can contribute to liver-related fatigue.
Monica C. Skarulis, M.D. (staff clinician, Clinical Endocrine Section, and acting director of the Metabolic Clinical Research Unit, NIDDK), studies the effects of excess weight and obesity on health. She is conducting a study that describes the physical and behavioral traits of overweight and obese adults and characterizes the hormones, metabolism, food preferences, fitness and physical-activity levels, sleep patterns, and thought processes in people with and without weight problems.
Kong Chen, Ph.D. (investigator and director, Human Energy and Body Weight Regulation Core, NIDDK), focuses on human energy metabolism as it relates to health and disease. His group has developed several whole-room indirect calorimeters, also called respiration or metabolic chambers, which are used to measure the rate of energy expenditure day and night (including during sleep) on a minute-by-minute basis and substrate oxidation for several hours or for several days.
NATIONAL INSTITUTE OF ENVIRONMENTAL HEALTH SCIENCES
Serena Dudek, Ph.D. (senior investigator, Neurobiology Laboratory/Synaptic and Developmental Plasticity Group, NIEHS), doesn’t do sleep studies per se, but discovered that caffeine, at levels similar to those consumed by humans, strongly enhanced synaptic responses in rats in the CA2 region of hippocampus, an area of the brain known for its role in learning and memory. The CA2 region could, therefore, be a potential target for drug development to combat symptoms of fatigue due to sleep deprivation and depression as well as sleep disturbances in neurodevelopmental disorders, such as autism. To read more, go to https://irp.nih.gov/catalyst/v20i1/coffee-may-boost-learning-potential.
Honglei Chen, M.D., Ph.D. (senior investigator, Epidemiology Branch; head, Aging and Neuroepidemiology Group, NIEHS), hopes to characterize populations that are at risk for Parkinson disease and to understand its etiology. He is using data from several large population studies to track participants with clusters of prodromal nonmotor symptoms such as loss of smell, constipation, and sleep disturbances. In 2011, he reported that longer daytime napping was associated with a higher risk for Parkinson disease. In the future, he plans to evaluate sleep disturbances as early nonmotor symptoms for Parkinson disease.
Janet Hall, M.D. (incoming clinical director, NIEHS), is doing research on neuroendocrine interactions underlying normal human reproduction and the changes that occur both with aging and in clinical disorders of ovulation. In collaboration with a Harvard Medical School investigator, she is studying the effects of sleep and nutrition on pubertal progression in girls.
NATIONAL INSTITUTE OF MENTAL HEALTH
Carolyn Beebe-Smith, Ph.D. (senior investigator and chief, Section on Neuroadaptation and Protein Metabolism, NIMH). See interview.
Ashura Buckley, M.D. (staff clinician, Pediatric and Developmental Neuroscience Branch, NIMH), is collaborating with Susan Swedo, M.D., to investigate how abnormal sleep patterns may contribute to autism spectrum disorders (ASD). Buckley is conducting a randomized controlled trial to see whether donepezil, which has been shown to increase rapid-eye-movement sleep in children with ASD, can improve communication and social interaction skills in these children.
Kathleen Merikangas, Ph.D. (senior investigator and chief, Genetic Epidemiology Research Branch, NIMH), has devoted substantial effort to characterizing the prevalence and correlates of sleep patterns and disorders in both general population samples and a local community-based family cohort of more than 1,000 individuals. As part of an international collaborative effort on activity and mood disorders, her research group has developed a diagnostic interview for sleep patterns and problems (Sleep Med 15:530-535, 2014) and piloted the use of mobile technologies (activity monitors and electronic diaries) in their community family study to track sleep, eating, activity, and mood states in real time. Analyses of national data on adults have shown that poor sleep is associated with increased concentrations of inflammatory markers and poor physical and mental health, particularly cardiovascular disorders and migraine (Sleep 36:671-679, 2013). In large-scale population studies of adolescents in the United States, the research team has demonstrated that sleep duration and difficulties are associated with serious health consequences including depression, suicidality, and poor physical health (Cephalalgia 31:648-653, 2011; Am J Psychiatry 170:501-510, 2013; Sleep, in press). Merikangas’s team is now developing pilot interventions to reduce the impact of sleep problems on health and functional impairment.
Susan Swedo, M.D. (senior investigator, and chief, Pediatric and Developmental Neuroscience Branch, NIMH), and Ashura Buckley, M.D., are studying how abnormal sleep patterns may contribute to autism spectrum disorders (ASD). Some of these children spend very little time in the rapid eye movement (REM) stage of sleep, and researchers want to know whether REM sleep abnormalities contribute to the social and communication deficits seen in ASD. Swedo is conducting clinical and immunological investigations of subtypes of autism, including measuring brain waves during sleep. To read more about their work, go to https://irp.nih.gov/catalyst/v20i3/piecing-together-the-autism-puzzle.
Audrey E. Thurm, Ph.D. (staff scientist, Pediatrics and Neuroscience Branch, NIMH), and Ashura Buckley, M.D., are studying autism spectrum disorders (ASD) and conducting a pilot study on the markers of ASD in at-risk toddlers. The study includes an overnight sleep study, which involves an electroencephalogram test to measure brain electrical activity, and a magnetic resonance imaging scan.
Thomas Wehr, M.D. (scientist emeritus, NIMH; former chief of NIHM’s Clinical Psychobiology Branch), reported in a 1992 study that humans would revert back to a pre-industrial era two-shift pattern of sleep (two four-hour shifts, separated by one to three hours of quiet wakefulness) if they were not exposed to artificial lighting. His findings indicate that waking up in the middle of the night may be a normal physiological event, not a sleep disorder; they have implications for how sleep is regulated by two circadian oscillators that are separately entrained to dusk and dawn (J Physiol 535:937–951, 2001).
Carlos A. Zarate, M.D. (senior investigator and chief, Section on the Neurobiology and Treatment of Mood Disorders, Experimental Therapeutics and Pathophysiology Branch, NIMH), is conducting a clinical trial that is examining whether riluzole (an FDA-approved drug for treating amyotrophic lateral sclerosis, also known as ALS or Lou Gehrig’s disease) can improve symptoms of depression such as depressed mood, psychomotor retardation, and excessive sleeping in patients with bipolar disorder. In one of his studies, he found that sleep efficiency in the middle of the night (measured with polysomnography) is strongly associated with suicidal ideation and so could be a biomarker of suicidal thoughts. Among his other studies was the first study to describe the effects of ketamine infusion on electroencephalography sleep slow waves and improvement in depressive symptoms in patients with treatment-resistant major depressive disorder (Int J Neuropsychopharmacol 16:301–311, 2013).
NATIONAL INSTITUTE OF NURSING RESEARCH
Jessica Gill, R.N. Ph.D. (Lasker Clinical Research Scholar, Tissue Injury Branch, NINR), is conducting a clinical trial to investigate the biological and neuronal mechanisms, including sleep disturbances, of post-traumatic stress disorder, depression, and post-concussive syndrome after a traumatic brain injury (TBI). She hopes to gain a better understanding of how sleep contributes to the psychiatric and neurological recovery from a TBI, because current treatments for psychiatric risk are often ineffective. To read more about her work, go to https://irp.nih.gov/catalyst/v21i1/nih-lasker-scholars.
Leorey N. Saligan, Ph.D., R.N., C.R.N.P. (investigator and chief, Symptom Management Branch, NINR), has been investigating the nature and causes of fatigue in cancer patients and in healthy individuals. He and his team have identified genes that can predict fatigue risk for patients receiving cancer therapy. They plan to do a secondary analysis of the genomic data to see whether there are shared functional networks among common differentially expressed genes in cancer patients with sleep disturbances and fatigue. To read more about his work, go to https://irp.nih.gov/our-research/research-in-action/energized-by-fatigue.
INTERVIEW WITH MICHAEL J. TWERY, PH.D.

Michael J. Twery, the diirector of NIH’s National Center on Sleep Disorders Research (NCSDR), has led the NIH’s sleep and respiratory neurobiology scientific research group since 1996, and has served as director of the NCSDR since January 2006. In these roles, he oversees the support of research and research training related to sleep disordered breathing, the fundamental functions of sleep and circadian rhythms, and sleep disorder epidemiology and clinical trials. The NCSDR also stewards several forums that facilitate the coordination of sleep research across the NIH, other federal agencies and outside organizations, including the Sleep Disorders Research Advisory Board and a Trans-NIH Sleep Research Coordinating Committee. Earlier in his career, Twery was a senior staff fellow and studied Parkinson Disease, at the National Institute of Neurological Disorders and Stroke. Following are edited excerpts of his interview with the NIH Catalyst.
What happens when we don’t get enough sleep?
Lack of sleep can affect our clarity of thought, emotions, and how we perceive or interpret emotions in others. It can also affect our immune system, growth and stress hormones, and even our blood pressure. People are familiar with the need to tune a gasoline engine in a car. If the timing is off, even a little bit, the engine operates inefficiently, burns fuel inefficiently, and gets fewer miles per gallon. By analogy, this is similar to what happens to our body chemistry when we don’t follow a regular schedule of sleep. To keep “tuned up,” we need to sleep at the right time and get enough good-quality sleep. And just like an engine that needs a tune-up, your sleep-deprived body will keep working, maybe for years. You might be a little harder to start and you don’t get as good “mileage.” In the end, an uncared-for engine will die an early death than an engine that’s been taken care of…
How much sleep do we need?
There’s a lot of confusion and controversy. You may feel you are getting enough sleep when you don’t feel sleepy. But what might help prevent you from feeling sleepy—like a three-hour nap—doesn’t contribute a lot to long-term health because it doesn’t support the timing of our 24-hour circadian rhythms that regulate our internal clock. Generally, an adult needs seven to eight hours of sleep a night.
How does sleep affect health?
Only in recent years, we have begun to understood that sleep is one of the factors, like diet and exercise, on which our long-term health depends. Controlling the duration, timing, and quality of sleep to achieve best health outcomes is not going to correct a heart valve or decades of atherosclerosis, but it could help us maintain our health longer.
What do you think about the various mobile devices that supposedly measure sleep?
Some researchers may consider them toys because they aren’t as precise as research-validated devices. But they may help guide individuals’ health practices and give them access to information that is useful and that motivates them to pursue a healthy lifestyle.
What is microsleep and how do we prevent it?
By the time you start to experience and recognize sleepiness, it’s very advanced. As the day progresses and the pressure to sleep increases, and the signals to remain awake decline, there’s a period of transition. Our brain can begin to sleep (“switch off”) for microseconds. That’s called microsleep, brief moments of sleep that occur when you are normally awake. A microsleep lasting one or two seconds, if you’re driving, could result in a serious accident. The best defense is to get enough sleep at the right time, and good quality sleep, as a starting point to safeguard against microsleep.
Can you comment on some of the research going on at NIH?
Some early NIH sleep research included neuroendocrinology and the rhythms of hormones; the brain body connection and the idea of circadian sleep biology; and segmented sleep. Elucidating the nuts and bolts of sleep and sleep disorders is the cutting edge for much of the research that is going on today. Susan Harbison (NHLBI), for instance, is using state-of the-art techniques in fruit flies to break down sleep into measurable parts. Flies have a short life span, so if we manipulate them and deprive them of sleep, we can measure the effects on them very quickly and get a clue about how it might work in people. Kathleen Merikangas (NIMH) has published some interesting work [that shows that adolescents universally have less than the recommended amount of sleep and that prolonged fatigue is associated with significant disability]. NIDDK researchers are looking at circadian rhythms in the liver. This organ has one of the most robust circadian rhythms. You can put chunks of liver in a dish and they will continue to oscillate all by themselves. That observation showed that it’s not a matter of night and day, but a genetic program that’s built into every cell of the body. Several intramural researchers are looking at melatonin, a hormone involved with sleep. Light, even a little bit of light, can suppress the normal secretion of melatonin. Melatonin levels normally peak just before we are supposed to be going to bed. We are particularly sensitive to blue light (such as from your computer screen), because it suppresses the secretion of melatonin. Research is underway to learn how suppressing melatonin may increase the risk of breast cancer and gastrointestinal cancer.
Was snoring always considered bad for you?
In 1990s, snoring was taken as a sign of peaceful rest. Today, snoring can mean you’re struggling to breathe and in some people that’s a problem. Peaceful, quiet snoring might be okay, but loud, chronic snoring—called sleep apnea—is not a good sign. If not treated, sleep apnea can lead to other problems such as high blood pressure, stroke, or memory loss.
What effect does sleep or time of day have on when medications are administered?
Many years ago NCI funded a landmark study that showed that regulating the time of day of taking tamoxifen made a difference in the five-year survival of people with certain types of breast cancer. This research suggests some questions that could be explored: Can we improve efficacy of drugs by administering them at different times of day so they work in harmony with body? Because hormones vary at different times of day, are there periods of greater vulnerability to tissue injury or stress? Answers to these questions may increase our understanding of how we can modify risk. Another extramural study suggested that well-rested people had a 20-30 percent healthier response than sleep-deprived people to the influenza vaccine. Research is needed to learn whether people should be advised to to get a good night’s sleep before getting the flu vaccine.
The National Center on Sleep Disorders Research, located in the National Heart, Lung, and Blood Institute, oversees the support of research and research training related to sleep disorders and stewards several forums that facilitate the coordination of sleep research across NIH, other federal agencies, and outside organizations. For more information, visit http://www.nhlbi.nih.gov/about/org/ncsdr.
This page was last updated on Tuesday, April 26, 2022
