Back Page: Catalytic Research
NICHD: First 3-D Structure of DHHC Enzymes
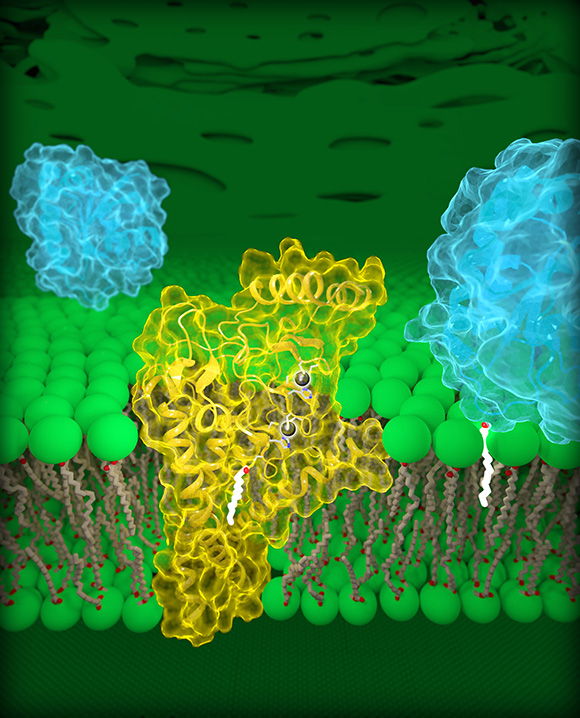
CREDIT: JEREMY SWAN, NICHD
The first three-dimensional structure of DHHC proteins—proteins that contain a 50-amino-acid chain called the DCCT domain, act as enzymes, and are involved in many cellular processes, including cancer—explains how they function and may offer a blueprint for designing therapeutic drugs. Researchers from the Eunice Kennedy Shriver National Institute of Child Health and Human Development led a study in which they proposed blocking DHHC-domain activity to boost the effectiveness of first-line treatments against common forms of lung and breast cancer. There are currently no licensed drugs that target specific DHHC enzymes. Shown: Molecular view of DHHC palmitoyltransferases. Human DHHC20 (yellow) is embedded in the Golgi membrane (green), a compartment located inside cells. DHHC20 attaches a fatty-acid chain (white) to a target protein (blue, foreground), which anchors the protein to the Golgi membrane.
DHHC enzymes, also called palmitoyltransferases, modify other proteins by attaching to them a chain of lipids, or fatty acids, of varying lengths. This modification, called palmitoylation, can change many properties of a target protein, such as its structure, function, and location within a cell. Researchers estimate that nearly 1,000 human proteins undergo palmitoylation, including epidermal growth-factor receptors (EGFRs). A well-known EGFR is human EGFR 2, or HER2, which is overactivated in aggressive forms of breast cancer. EGFRs can also be overactivated in colon cancer and non-small-cell lung cancer, the most common type of lung cancer.
The researchers identified a structural component of DHHC20 that influences the length of its lipid chain. Mutations that altered the relative size of this component caused DHHC20 (zinc finger DHHC-type containing 20) to use shorter or longer lipid chains, which presumably changes the effects of palmitoylation on a target protein. The researchers propose that the structure of this site explains why different DHHC enzymes use certain lipid chains to modify the functions of other proteins. It also offers insight on how multiple enzymes work together in states of health and disease. (NIH authors: M.S. Rana, P. Kumar, C.-J. Lee, R. Verardi, and A. Banerjee, Science 359:eaao6326, 2018; DOI:10.1126/science.aao6326)
This page was last updated on Friday, April 8, 2022
