Research Briefs
NCATS, NHLBI: INTERNATIONAL SCIENTIFIC TEAMS FIND POTENTIAL APPROACH AGAINST PARASITES
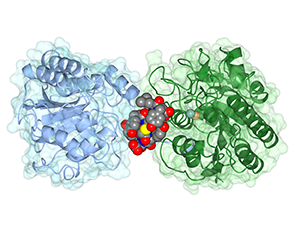
CREDIT: JIM INGLESE, NCATS
NCATS, NHLBI: The cyclic peptide ipglycermide binds to and blocks the activity of a cofactor-independent phosphoglycerate mutase (iPGM) enzyme, which is essential for glycolysis in disease-causing parasites and bacteria.
Parasitic nematode infections cause many devastating infectious diseases around the world. Researchers from NCATS and NHLBI and the University of Tokyo identified the first inhibitor of an enzyme long thought to be a potential drug target for fighting disease-causing parasites and bacteria. After sorting through more than 1 trillion small-protein fragments (cyclic peptides), the scientists found two potential inhibitors of the enzyme. The target enzyme—cofactor-independent phosphoglycerate mutase (iPGM)—exists in both parasites and bacteria and is essential for glycolysis. The team collaborated with structural biologists at the University of Kansas (Lawrence, Kan.) to determine the structure of the iPGM-cyclic peptide arrangement and how it prevented the enzyme from working properly. Next, the researchers will look for ways for cyclic peptides to enter cells. This study could inform developments of broad-spectrum antiparasitic and antibacterial agents. (NIH authors: P. Dranchak, R. MacArthur, N.J. Baird, and J. Inglese, Nat Commun 8:14932, 2017; DOI:10.1038/ncomms14932; http://www.readcube.com/articles/10.1038/ncomms14932)
NHGRI: SELFIES COULD HELP DIAGNOSE A RARE GENETIC DISEASE
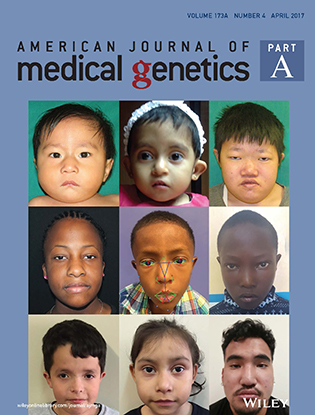
Taking selfies may one day help physicians diagnose genetic diseases such as DiGeorge syndrome, which is also known as 22q11.2 deletion syndrome and is caused by a defect in chromosome 22. Those with the syndrome exhibit certain facial features—such as an underdeveloped chin, low-set ears, wide-set eyes, or a narrow groove in the upper lip—and may have congenital heart disease; poor immune-system function; and delays in growth, development, and speech. The disorder is commonly underdiagnosed, especially in diverse populations. NHGRI scientists used digital facial-detection analysis on 101 photos—from consenting individuals with the syndrome—from 11 countries to determine 126 distinct geometric and textural facial features that serve as markers of the syndrome. The facial-recognition technology accurately diagnosed the syndrome 96.6 percent of the time across diverse populations. The prevalence of smartphones in areas of the world that lack molecular- and cytogenetic-testing facilities means that facial diagnostic capabilities of handheld devices can enhance early detection and treatment of 22q11.2. (NIH authors: P. Kruszka, Y.A. Addissie, J. Duncan, A.A. Adeyemo, and M. Muenke, Am J Med Genet 173:879–888, 2017; http://onlinelibrary.wiley.com/doi/10.1002/ajmg.a.38199/full)
NIBIB: NOVEL RADIOTRACER MAY IDENTIFY PROSTATE CANCER
There is no single imaging technique or test for diagnosing, monitoring, and staging prostate cancer. NIH researchers, however, are taking steps to develop a single method that, in one scan, can identify the cancer in its early stages as well as after metastasis. Using a novel radiotracer that recognizes two biomarkers normally found in prostate cancer–gastrin-releasing peptide receptor and integrin alpha-V beta-3–the researchers successfully identified 75 percent of primary prostate tumors and 100 percent of metastatic lymph nodes and bone lesions in the study cohort (five healthy volunteers and 13 patients with various stages of prostate cancer). The probe (gallium-68–bombesin–arginylglycylaspartic acid) was well tolerated and outperformed prostate biopsy, magnetic resonance imaging, and positron-emission tomography (PET) using the PET tracer 18F-FDG in detecting prostate cancer and metastatic lesions. Combined with other imaging tools, this new technique could provide tumor-staging information and monitoring response to therapy and could serve as a guide for delivering internal radiation therapy. Larger-scale clinical studies are needed to confirm the findings. (NIH authors: J. Zhang, G. Niu, L. Lang, X. Yan, X. Chen, J Nucl Med 58:228–234, 2017; http://jnm.snmjournals.org/content/58/2/228.full.pdf)
NIAMS, NHLBI, OD: TREATING LUPUS WITH TOFACITINIB
Tofacitinib, a drug already approved for the treatment of rheumatoid arthritis, prevented the onset of lupus in lupus-prone mice, NIH researchers found. Lupus is a chronic autoimmune disease that causes widespread inflammation and eventually leads to cardiovascular and renal damage. It is thought that high concentrations of cytokines, especially interferons, are associated with the progression of the disease. Tofacitinib, which was discovered and developed at NIH, blocks the inflammatory response pathway Janus kinase–signal transducers and activators of transcription. When administered to mice that had already developed lupus, tofacitinib reversed kidney damage, skin inflammation, and blood-vessel abnormalities. In mice that had not yet developed lupus, the drug prevented onset of the disease. Further studies are needed to determine the efficacy and safety of using tofacitinib to control lupus symptoms in humans. (NIH authors: Y. Furumoto, C.K. Smith, L. Blanco, W. Zhao, S.R. Brooks, S.G. Thacker, A. Zarzour, G. Sciumè, W.L. Tsai, A.M. Trier, L. Nunez, L. Mast, V. Hoffmann, A.T. Remaley, J.J. O’Shea, M.J. Kaplan, and M. Gadina, Arthritis Rheumatol 69:148–160, 2017; https://www.ncbi.nlm.nih.gov/pubmed/27429362)
NCI: THYROID CANCER ON THE RISE
The incidence of thyroid cancer has increased by 211 percent in the United States over the past 40 years, according to an NCI-led study. Researchers at NCI and Duke University Medical Center (Durham, N.C.) analyzed data from more than 77,000 thyroid-cancer patients diagnosed during 1974–2013 and from more than 2,000 thyroid-cancer deaths during 1994–2013. Although some investigators have thought that the increase was due to improvements in diagnostic technology that picked up small tumors that would have never required treatment, the current study challenges that notion. Additional research is needed, however, to determine whether environmental factors—such as endocrine-disrupting chemicals or changes in obesity and smoking prevalence—may be associated with the increase. (NIH authors: H. Lim, S.S. Devesa, D. Check, and C.M. Kitahara, JAMA 317:1338–1348, 2017; http://jamanetwork.com/journals/jama/fullarticle/2613728)
NIDCD: GENE THERAPY MAY BE EFFECTIVE IN TREATING HEARING LOSS AND DIZZINESS DISORDER
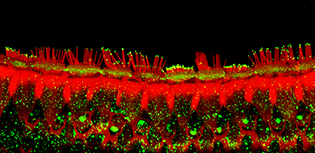
CREDIT: NIDCD
NIDCD: Gene therapy with Whrn restores stereocilia bundles (red) in the inner ear of a Whrn mutant mouse to normal length and position. Even after four months, stereocilia continue to produce whirlin protein (green), helping to maintain the structure and function of the stereocilia and improving the mouse’s ability to hear and maintain balance.
Using gene therapy, scientists from NIDCD and Johns Hopkins School of Medicine (Baltimore) have corrected defective structures in the inner ears of newborn mice, with long-term benefits. The study is one of the first to use gene therapy successfully to improve hearing and restore balance in mice with a type of inherited deafness, called Usher syndrome, also found in people. The findings support growing evidence that gene therapy could be an effective treatment for inherited types of hearing loss, deafness, and balance disorders. The experimental gene therapy used in the study targets hair cells, which are sensory cells in the inner ear. These cells have hair-like projections called stereocilia that help detect and process incoming sound and information about movement to guide a person’s sense of balance. Stereocilia need the protein whirlin to fully grow and function properly. In one type of Usher syndrome, the gene that encodes whirlin, Whrn, is mutated. This causes the stereocilia to be abnormally short and disorganized, making them dysfunctional. As a result, the sensory hair cells cannot detect sound and information about movement to guide the sense of balance, leading to dizziness and to hearing loss or deafness. Read full article on NIDCD webpage at https://www.nidcd.nih.gov/news/2017/gene-therapy-usher-syndrome. (NIH authors: K. Isgrig, J.W. Shteamer, I.A. Belyantseva, M.C. Drummond, T.S. Fitzgerald, A.J. Griffith, T.B. Friedman, L.L. Cunningham, and W.W. Chien, Mol Ther 25:780–791, 2017, bit.ly/2q7m3tc)
NIAMS: MECHANISMS TO IMPROVE NUCLEAR REPROGRAMMING
NIAMS researchers conducted high-throughput RNA sequencing of transplanted oocytes to uncover the mechanisms that cause some genes to resist reactivation. The findings will help improve the success of nuclear reprogramming. (NIH authors: S. Wang and V. Sartorelli, Molec Cell 65:873–884, 2017; http://www.cell.com/molecular-cell/fulltext/S1097-2765(17)30056-4)
NICHD: SCREENING TESTS FAIL TO PREDICT PRETERM BIRTH IN FIRST-TIME PREGNANCIES
Two screening methods that once seemed promising for predicting premature deliveries in first-time pregnancies may not be so helpful after all. NICHD researchers screened more than 9,000 women throughout their pregnancies by evaluating routine ultrasound examination of the uterine cervix (a short cervix early in pregnancy could be a warning sign of preterm birth) and testing for fetal fibronectin (some studies have suggested that the presence of this gluelike protein in the vagina early in pregnancy could signal early labor). But the screenings failed to identify most of the women who would go on to give birth prematurely. The researchers concluded that, alone or together, the methods did not identify enough preterm births to support using them for routine screening of first-time pregnancies. (NIH author: U.M. Reddy, JAMA 317:1–10, 2017; http://jamanetwork.com/journals/jama/fullarticle/2610337)
NHGRI: AFRICAN-SPECIFIC GENOMIC VARIANT ASSOCIATED WITH OBESITY
An international team of researchers, including some from NHGRI, has conducted the first study of its kind to look at the genomic underpinnings of obesity in continental Africans and African-Americans. They discovered that approximately 1 percent of West Africans, African-Americans, and others of African ancestry carry a genomic variant that increases their risk of obesity, a finding that provides insight into why there are obesity clusters in families. The burden of obesity is, however, not the same across U.S. ethnic groups, with African-Americans having the highest age-adjusted rates of obesity. This study is the first to use a genome-wide association study to investigate the genomic basis of obesity in continental Africans. The researchers plan to replicate these findings in more populations and conduct experiments using cell lines and model organisms such as zebrafish (Danio rerio) to identify the role of variants in the gene SEMA4D in obesity and obesity-related traits. (The SEMA4D gene plays a role in cell signaling, the immune response, and bone formation.) Available data show that the newly identified genomic variant overlaps a region of DNA called an “enhancer” that can be activated to increase the work of a particular gene. (NIH authors: G. Chen, A.P. Doumatey, J. Zhou, L. Lei, A.R. Bentley, F. Tekola-Ayele, S.N. Adebamowo, J.L. Baker, D. Shriner, A.A. Adeyemo, and C.N. Rotimi1, Obesity 25:794–800, 2017; http://onlinelibrary.wiley.com/doi/10.1002/oby.21804/full)
NICHD: EXTREME TEMPERATURES MAY INCREASE RISK FOR LOW BIRTH WEIGHT
Experiencing extreme hot or cold temperatures during pregnancy may increase the risk that infants born at term will be of low birth weight, according to an NICHD study of U.S. women. The authors found that exposure to atypically cold temperature during the entire pregnancy, or just during the second trimester and third trimester, increased the risk for low birth weight. Exposure to atypically hot temperatures during the whole pregnancy, or during the third trimester, also increased this risk. The odds for low term birth weight were highest when the woman was exposed to extreme temperatures during her whole pregnancy. In the current study, NICHD researchers linked medical records from 223,375 births at 12 U.S. clinical centers to hourly temperature records for the region surrounding each center. Infants born at term to mothers exposed to cold during either the second or third trimester were 18 to 21 percent more likely to be of low birth weight. When exposed to extended cold over the entire pregnancy, term infants were 257 percent more likely to be of low birth weight. Term infants exposed to heat in the third trimester were 31 percent more likely to be of low birth weight, and infants born after the entire pregnancy occurred during atypically hot temperatures were 249 percent as likely to be of low birth weight. Researchers do not know why exposure to high or low temperatures would affect birth weight. The authors noted that previous studies have suggested that heat exposure could affect birth weight by increasing oxidative stress and inflammation. It is also possible that temperature extremes could reduce blood flow to the uterus, depriving the infant of oxygen and nutrients and hindering the placenta’s ability to remove fetal wastes. (NIH authors: S. Ha, Y. Zhu, D. Liu, P. Mendola, Environ Res 155:394–400, 2017; http://www.sciencedirect.com/science/article/pii/S0013935116307332)
NICHD, NHGRI: ORIGINS OF BLOOD–BRAIN BARRIER “SENTRY CELLS”
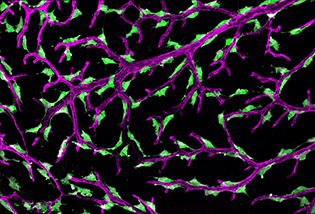
CREDIT: NICHD
An adult zebrafish brain showing fluorescent granular perithelial cells (green) atop blood vessels (purple). Top: An image of an adult zebrafish brain showing fluorescent granular perithelial cells (green) atop blood vessels (purple). Bottom: A close-up of fluorescent granular perithelial cells atop blood vessels in a zebrafish brain.
A study conducted by researchers at NIH and the Japanese National Institute of Genetics may have implications for understanding age-related decline in brain functioning and how human immunodeficiency virus (HIV) infects brain cells. The scientists determined that in embryonic zebrafish (Danio rerio), a population of cells that protects the brain against diseases and harmful substances are not immune cells, as had previously been thought, but instead likely arise from the lining of the circulatory system. In the current study, the researchers showed that fluorescent granular perithelial cells (FGPs) are present on the surface of the zebrafish brain and that these blood vessel–associated FGPs do not arise from the immune system, but from endothelial cells themselves.
FGPs are thought to be important in a variety of human brain disorders and conditions. These cells appear to be a major entry point for HIV infection of the brain. Age-related decline in cognitive function is associated with a decline in the scavenging function of FGPs. The researchers hope to conduct further studies of how FGPs interact with blood vessels and the blood–brain barrier. The researchers noted that, unlike mammals, embryonic zebrafish can be observed under a microscope as they develop, providing an easy means for studying the role of FGPs in protecting the brain. (NIH authors: M.V. Galanternik, D. Castranova, A.V. Gore, N.H. Blewett, H.M. Jung, A.N. Stratman, M.R. Kirby, J. Iben, M.F. Miller, R.J. Maraia, and B.M. Weinstein, eLife 2017;10.7554/eLife.24369; DOI:http://dx.doi.org/10.7554/eLife.24369; https://elife.elifesciences.org/content/6/e24369)
NIAID, CC: NIH STUDY OF EBOLA PATIENT TRACES DISEASE PROGRESSION AND RECOVERY
CREDIT: NIAID
Ebola virus isolated in November 2014 from patient blood samples obtained in Mali. The virus was isolated on Vero cells.
Analysis of daily gene activation in a patient with severe Ebola virus disease cared for at the NIH Clinical Center in 2015 found changes in antiviral and immune-response genes that pinpointed key transition points in the response to infection. A NIAID-led study found that the changes included a marked decline in antiviral responses that correlated with clearance of virus from white blood cells. The analysis also showed that the preponderance of host responses shifted rapidly from activation of genes involved in cell damage and inflammation toward those linked to promotion of cellular and organ repair. This pivot came before the first signs of clinical improvement in the patient, who was admitted to the NIH Clinical Center on day 7 of illness and remained at the hospital for 26 days. Although the study represents only one patient, it provides unprecedented detail on the host response to Ebola virus disease and may inform the development of therapeutics. It may also lead to better prognostic criteria to enable clinicians to tailor the treatment of patients with Ebola virus disease in ways that can best promote recovery. (NIH authors: J.C. Kash, J. Kindrachuk, K.B. Janosko, R.D. Adams, M.J. Memoli, R.T. Davey Jr, D.S. Chertow, and J.K. Taubenberger, Sci Transl Med 9:eaai9321, 2017; DOI:10.1126/scitranslmed.aai9321; http://stm.sciencemag.org/content/9/385/eaai9321)
NIAID: NIH SCIENTISTS ADVANCE UNDERSTANDING OF HERPESVIRUS INFECTION
The World Health Organization estimates that about 500 million people are infected with Herpes simplex virus (HSV-2) and more than 3.7 billion people under the age of 50 are infected with HSV-1. These viruses cause human diseases ranging from oral cold sores to genital lesions to serious eye conditions that can lead to blindness. In infants, HSV can cause neurological and developmental problems. People infected with HSV also have an enhanced risk of acquiring or transmitting human immunodeficiency virus.
HSV infections last a lifetime. Once a person has been infected, the virus can remain dormant (latent) for years before periodically reactivating to cause recurrent disease. This poorly understood cycle has frustrated scientists for years. Now, NIAID scientists, in collaboration with scientists from Princeton (Princeton, N.J.), have identified a set of protein complexes that are recruited to viral genes and stimulate both initial infection and reactivation from latency. Environmental stresses known to regulate these proteins also induce reactivation. (NIH authors: R. Alfonso-Dunn, A-M.W. Turner, J.H. Arbuckle, and T.M. Kristie, Cell Host Microbe 21:507–517,e5, 2017; DOI:10.1016/j.chom.2017.03.007; http://www.sciencedirect.com/science/article/pii/S1931312817301178)
This page was last updated on Saturday, January 18, 2025
