Research Festival: Selected Symposia 2015
Battling the Bugs: But Maybe NOT with Antibiotics
Although it sounds unbelievable, each of us carries around 10 times as many microbes as we have human cells. Together, these microbes form our very own microbiome that is intricately linked to our personal health. The growing problem of drug resistance, however, is among the greatest threats to public health facing us today.
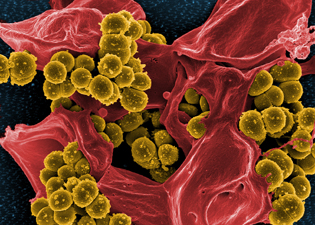
IMAGE BY FRANK DELEO, NIAID
Scanning electron micrograph of methicillin-resistant Staphylococcus aureus and a dead human neutrophil
“Antibiotic resistance in bacteria is a problem worldwide,” said Karen Frank, workshop chair and chief of the Clinical Center’s Microbiology Section. She went on to explain that tracking drug-resistant bacteria from hospitals and patient isolates and using advanced genomic tools such as next-generation sequencing to analyze them can help researchers identify the mechanisms bacteria use to spread resistance. It will eventually help in better treatment and disease prevention.
According to Michael Otto (National Institute of Allergy and Infectious Diseases), Staphylococcus aureus is the most common pathogen in hospital-acquired infections and the “number-one killer.” Community-associated infections are on the rise worldwide, and they “can do a lot of nasty things,” he added. The objective of his research is to find effective virulence-targeted therapeutic drugs that can interfere with the antibiotic-resistance process.
Wei Sun, a staff scientist from the National Center for Advancing Translational Sciences, expressed similar concerns and explained how high-throughput screening can help identify different drug-combination regimens for multidrug-resistant bacterial infections.
Our skin is an important immune barrier, and “it is very complex with regards to what microbial communities are present on the skin surface,” said Heidi H. Kong from the National Cancer Institute (NCI). The skin microbiota can also be site-specific and differ between healthy skin and skin afflicted with such conditions as atopic dermatitis. She also mentioned that just as host factors influence the skin microbiota, skin microbiota can influence host skin. One of her research goals is to determine whether microbiota can be sufficiently modified to promote human health in the future.
FARE Award winner Majda Haznadar (NCI) presented her research on lung cancer metabolomics. Because lung cancer is commonly diagnosed at a late stage using invasive techniques, efficient and robust risk biomarkers that can detect the disease noninvasively at an early stage should greatly improve disease detection and diagnosis.
The intramural research program is continuing to advance the study of the interconnected realms of microbiome and drug resistance by building on the program’s complementary strengths in immunology, immunotherapy, microbial and human genomics, cohort studies, animal model systems, and access to well-defined patient populations in the Clinical Center.
The “Microbiome and Drug Resistance” session was chaired by Karen Frank (CC).
Vaccines Workshop: Challenges and Solutions in Vaccine Design
Vaccines, the topic of a workshop at the 2015 NIH Research Festival, are one of the most effective preventative measures we have to control infectious diseases. Yet despite growing numbers of effective vaccines, there are still many pathogens we don’t yet know how to tackle.
When designing a vaccine for malaria, as Patrick Duffy (National Institute of Allergy and Infectious Diseases, NIAID) is attempting to do, which life stage do you choose? Do you vaccinate to prevent infection in people? Or do you vaccinate to reduce the amount of malaria in mosquitos to reduce the risk of their transferring the disease?
Or, when attempting to design a vaccine for respiratory syncytial virus (RSV), a disease that can be deadly in newborns, how do you prevent infection in patients who can’t themselves be vaccinated? Barney Graham (NIAID’s Vaccine Research Center, VRC) discussed this quandary, as well as new insights gained from novel structures of the RSV F protein, as an example of structure-based vaccine design.
Kanta Subbarao (NIAID) and FARE Award winner John Gallagher (NIAID) delved into the ongoing challenges of increasing the breadth of protection conferred by existing influenza vaccines and of producing an effective, universal vaccine for influenza. The most conserved piece of the infection machinery on the outside of the virus is the “stem” of the influenza hemagglutinin protein. New vaccine designs direct the immune response away from the variable “head” region and toward this conserved “stem“ region.
Stephen Whitehead (NIAID) discussed the challenges of creating a vaccine against dengue virus. Although the virus usually causes mild disease, it can sometimes cause severe disease when a person is infected sequentially with two or more different serotypes. To prevent the severe form of the disease, it was necessary to design a vaccine that would protect equally against all four serotypes at once.
And then there are those diseases for which just stimulating antibodies is not enough. In her efforts to design a vaccine for Ebola virus, Nancy Sullivan (NIAID’s VRC) found that although antibodies often correlated with protection, in and of themselves the antibodies couldn’t neutralize the virus. Instead, the critical element for creating protection against the disease was picking a vector that stimulated CD8+ T-cell responses.
“The perception is, the [Ebola] outbreak happened; scientists got really busy, [and] made a vaccine almost instantly,” said Sullivan. But the vaccine had already been in the works. Creating vaccines for tricky targets is a long, difficult process, building on the knowledge acquired from each failure, as well as each new success, along the way.
The “Vaccines” workshop was chaired by Kanta Subbarao (NIAID).
Hitting the Brakes on Compulsive Behaviors
Like a car rolling downhill, activities such as eating ice cream or playing roulette can be hard to stop once they get started. When this inability to hit the brakes begins to have negative consequences, even everyday actions can be reclassified as compulsions. NIH researchers are eager to discover the brain systems that give rise to compulsive behaviors such as binge eating, substance abuse, and gambling addiction.
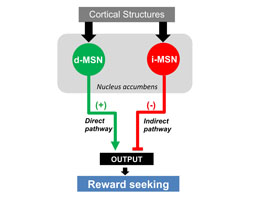
IMAGE BY VERONICA ALVAREZ, NIAAA
NIAAA’s Veronica Alvarez examined how cocaine changes two sets of neurons in the nucleus accumbens (NAc).
Addictive drugs alter a part of the brain called the nucleus accumbens (NAc), which contains “direct pathway” neurons that promote action and “indirect pathway” neurons that inhibit behavior. Despite changes in the NAc, not every person who uses drugs becomes addicted. NIAAA’s Veronica Alvarez discovered that the direct pathway becomes stronger in mice that are given cocaine, but the indirect pathway is also boosted in mice showing relatively few compulsive drug-seeking behaviors. Further experiments using optogenetic and chemogenetic methods confirmed that enhanced activity in the indirect pathway reduces the frequency of drug-seeking behaviors and may be protective against addiction.
Similarly, Lex Kravitz (NIDDK) found evidence that a high-fat diet causes loss of the dopamine receptors that inhibit the NAc’s indirect pathway, causing obese and formerly obese mice to move less. Using a designer receptor called kappa-opioid receptor DREADD (KORD) to restore inhibition of the indirect pathway rescued motor activity in obese mice.
FARE Award winner Nathan Marchant (NIDA) trained rats to self-administer alcohol in one environment (context A) but punished alcohol-seeking behavior in another environment (context B). The rats stopped consuming alcohol when in context B but resumed the behavior when placed back into context A, a process called context-induced relapse. However, when Marchant used muscimol to inactivate a brain structure called the ventral subiculum or used the KORD receptor to inhibit the ventral subiculum’s projections to the NAc, the rats showed significantly lower context-induced relapse to alcohol seeking, suggesting a role for the projection in context-induced relapse.
Related work by Bruce Hope (NIDA) showed that silencing the specific set of NAc neurons activated when genetically modified rats self-administer drugs in context A reduces context-dependent relapse. This finding provides evidence that associations between drugs and specific environments are encoded by particular groups of cells called “neuronal ensembles.”
Maximilian Muenke (NHGRI) discussed a study that examined the heredity of attention-deficit hyperactivity disorder (ADHD) in 18 families from Colombia, South America, in whom the condition is common. It was already known that ADHD has a genetic basis, but he linked it to some specific locations in the human genome that are related to the disorder and suggested that the disorder is often accompanied by an increased risk of other problems, including substance-abuse disorders.
Neurological disorders that lead to compulsive behaviors can substantially decrease quality of life. The NIH’s intramural research program hopes that understanding the neural circuits that produce such behaviors will help researchers produce therapies to combat those conditions.
The “Neuroscience and Compulsive Behavior” workshop chair was Veronica Alvarez (NIAAA).
Natural Products: The Future Therapeutics!
NIH scientists are discovering how natural products produced by bacteria, fungi, marine organisms, and plants can be used as powerful therapeutics. At the “Natural Products” workshop, several investigators discussed their work.
David Newman (National Cancer Institute, NCI), considered the world’s walking natural-products encyclopedia, described the evolution of the NCI’s Natural Products Repository (NPR), which was established in 1987. A few of the finest natural-product contributions to the field of medicine include the anticancer molecules doxorubicin, experimental drug PM00104 (Zalypsis), and paclitaxel (which was developed by NCI researchers).
In the war against malaria, National Center for Advancing Translational Research (NCATS) investigator James Inglese is using high-throughput screening techniques to examine thousands of extracts from marine sources to determine which of them are potent antimalarials.
FARE Award Winner Eugene Liu (National Institute of Allergy and Infectious Diseases) is trying to identify protein-specific features of the malaria parasite Plasmodium falciparum that are associated with variation in host immune responses.
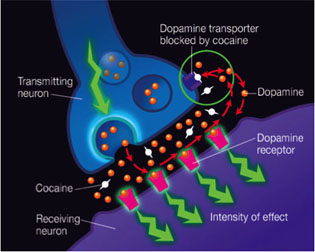
COURTESY OF NIDA
Cocaine, a natural product that comes from the coca leaf, is a local anesthetic that is highly addictive. Cocaine binds to the dopamine transporter and blocks dopamine reuptake, resulting in a superstimulation of dopamine receptors that produces the euphoric effects that can lead to addiction.
Amy Newman (National Institute on Drug Abuse) introduced the “dark side” of the natural products cocaine, heroin, and marijuana. She spoke about the need to develop addiction treatments and about her work targeting the dopamine D3 receptor (D3R) with novel D3R-selective antagonists and partial agonists. “We continue to tweak our template to optimize compounds as in vivo tools to test the D3R hypothesis,” she said.
The session was concluded by Barry O’Keefe (NCI), who foresees natural-products research becoming a priority over the next decade. He highlighted how the NCI Program for Natural Product Discovery (NPNPD) is benefitting from the NCI’s Center for Cancer Research collaboration with its Division of Cancer Treatment and Diagnosis. O’Keefe discussed approaches to enhancing the value of the NPR and for increasing the pace of discovery of bioactive molecules by the NPNPD. He also stressed the importance of applying bioinformatics as a tool for map analysis and prioritization of data. (https://dtp.cancer.gov/organization/npb/introduction.htm)
NIH’s vision for the future includes a comprehensive Natural Products Library with prefractionated compounds for modern high-throughput targeted screening technology and the creation of public databases and bioinformatics platforms to integrate source organisms and their biological activities and structural and genomic data.
“Nature has provided us with the largest arsenal of clinically used therapeutics outnumbering synthetic molecules by 2 to 1, said Carole Bewley (National Institute of Diabetes and Digestive and Kidney Diseases), who chaired the workshop. “I believe the future is bright for natural-products research and discoveries of new medicines.”
Note: Bewley and the other presenters at the workshop were thrilled when the 2015 Nobel Prize in Physiology and Medicine was awarded to three people who had developed anti-parasitic drugs from natural products (http://www.nature.com/news/anti-parasite-drugs-sweep-nobel-prize-in-medicine-2015-1.18507).
The “Natural Products” workshop was chaired by Carole Bewley (NIDDK).
Research Festival Posters and Bake-Off
The collaborative spirit was in full swing at the Research Festival’s poster sessions, one of which featured the work of scientific directors and institute directors. All the posters conveyed a snapshot of the promising research being done from bench to bedside at the NIH.

PHOTO BY ERNIE BRANSON
Catherine Bushnell, scientific director of the National Center for Complementary and Integrative Health, joined in the fun with NIH institute directors and scientific directors who exhibited posters and showed off their baking skills. Just behind her, facing the camera, is NINR Scientific Director Ann Cashion who took first place in the cooking contest.
In the institute and scientific directors’ poster session, two directors tied as first-place winners: Alan Koretsky (scientific director of the National Institute of Neurological Disorders and Stroke) in the basic-science category and Paul Sieving (director of the National Eye Institute) in the clinical sciences category.
Koretsky’s poster asked whether two brains are better than one. His lab found that rodent embryonic neural cortical precursor cells develop into new, healthy brainlike tissue when transplanted into the cerebral spinal fluid of adult rodents.
For his poster, Sieving asked whether gene-transfer therapy could improve the outcome of patients with the genetic form of retinoschisis, a disease in which a mutation in the gene RS1 on the X chromosome leads to macular degeneration in young males. His team determined the mechanisms underlying this vision loss, devised a treatment that delivers a healthy RS1 gene to the eye, and initiated clinical trails to determine the safety of this therapy.
The intramural directors put their baking skills to the test, too. There were many creative sweet treats, including tooth-shaped cookies from Martha Somerman, the director of the National Institute of Dental and Craniofacial Research. But it was a non-sweet treat that took first prize: a savory pesto spread prepared by the National Institute of Nursing Research’s scientific director Ann Cashion.
To read descriptions of all the posters displayed at the 2015 Research Festival, go to http://researchfestival.nih.gov/2015/posters.cgi.
To read summaries of the plenary sessions at the 2015 NIH Research Festival, go to https://irp.nih.gov/catalyst/v23i6/research-festival-selected-symposia-2015.
This page was last updated on Monday, April 25, 2022
