The SIG Beat: Flow Cytometry Winter Meeting
NEWS FROM AND ABOUT THE SCIENTIFIC INTEREST GROUPS
Highlights from the Flow Cytometry Interest Group 2019 Winter Meeting
BY RAFAEL VILLASMIL, NEI
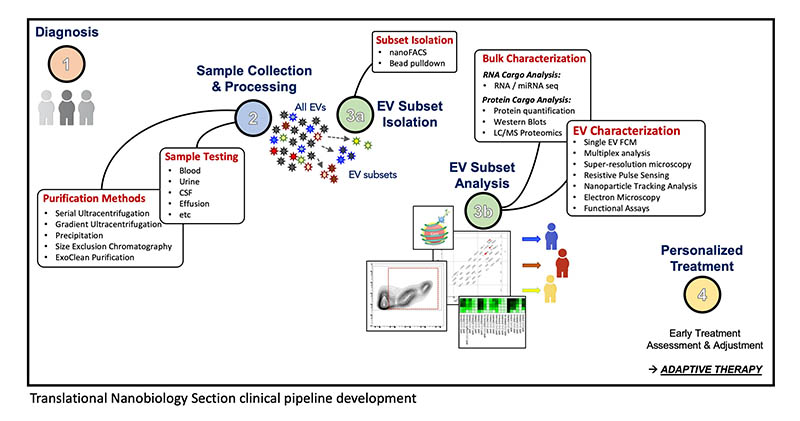
CREDIT: TRANSLATIONAL NANOBIOLOGY SECTION, NCI
An illustration depicting the Translational Nanobiology Section’s clinical pipeline, developed to analyze extracellular vesicles (EVs) as prognostic biomarkers of cancer therapies. A critical step is the use of flow cytometry to identify subsets of EVs and characterize them. Using EV data may lead to a better understanding of tumor and immune biology and could contribute to the development of personalized treatments.
Flow cytometry technology, which is constantly evolving, simultaneously analyzes physical and chemical characteristics of thousands of cells (such as blood or bone-marrow cells) as they move through fluid and are excited by a light source. By identifying and quantifying the cells of the immune system, flow-cytometry studies can characterize hematological malignancies and evaluate primary immune-deficiency disorders. Recent advances have enhanced the role of flow cytometry as an important method for characterizing immune function.
Some 150 attendees at the NIH Flow Cytometry Interest Group’s (FCIG’s) annual meeting (held on December 12, 2019, in Lipsett Amphitheater, Building 10) were eager to hear the latest research advances in the flow-cytometry field. “The meeting was a good educational event on the technological advances of [flow cytometry] and its applications, and an occasion for sound scientific exchange,” said Raul C. Braylan, senior clinician and chief of the Hematology Laboratory (NIH Clinical Center). It “offered a unique opportunity to learn technical advances of this very useful methodology and its importance in the evaluation and treatment of critical diseases such as immunodeficiencies and hematologic malignancy.”
Nine experts gave presentations including four NIH intramural researchers and five guests from biomedical companies.
Siak Jyh Kuen Jay, a visiting scholar at the National Eye Institute (NEI), presented his work on microorganisms that are that are living on the eye and described how he uses flow cytometry to monitor changes in the immune system. For example, cells collected from patients who have the autoinflammatory disease cryopyrin-associated periodic syndrome are prepared for cytometry and single-cell RNA sequencing. Jay discussed bioinformatic aspects of data analysis and plans for future use of oligonucleotide conjugates. Sergio Rosenzweig, chief of the Clinical Center’s Immunology Service, described how flow cytometry is used to diagnose immunodeficiencies by identifying and quantifying immune cells such as in blood, bone marrow, and other body fluids.
Flow cytometry is also used to characterize the quality of the chimeric antigen receptor T cells (CAR-T) and to measure the effectiveness of CAR-T therapy. National Cancer Institute (NCI) Investigator James Kochenderfer provided an update on CAR-T therapy clinical trials, which included a long-term follow-up of safety and efficacy for all subjects. Postdoctoral Fellow Joshua Welsh (Translational Nanobiology Section in NCI’s Laboratory of Pathology) talked about how flow cytometry is used to analyze extracellular vesicles (EVs), which are membrane-bound packages secreted by eukaryotic cells). Tumor cells, immune cells, and irradiated tissue release nanoscale EVs; using EV data may lead to a better understanding of tumor and immune biology.
Five guest speakers gave presentations on flow-cytometry instrumentation and software. Geoff Osborne (BD Biosciences) spoke about new instrumentation and latest version of the software FlowJo used to analyze flow-cytometry data. Giacomo Vacca (Kinetic River Corp.) presented innovative developments on time-resolved cytometry, a technique that measures the timing of fluorescence decay. The company received a Small Business Innovation Research grant from NIH and is developing two new instruments.
Joanne Lannigan (Flow Cytometry Support Services, LLC) described the spectral analysis technology in an instrument called the Aurora Cell Analyzer. James Freyer (BennuBio, Inc.) talked about an innovative fluidics system that generates several sample streams and processes samples at a rate 100 times as fast as typical cytometers can. Robert Thacker’s (Amnis Specialist Luminex Inc.) presentation featured new software that relies on machine learning and deep-learning neural networks in image cytometry to automatically classify cells.
The FCIG provides central information for basic and clinical investigators doing flow and image cytometry. All biomedical researchers with an interest in cytometry (regardless of institutional affiliation) are welcome. Visit https://oir.nih.gov/sigs/flow-cytometry-interest-group to learn more about this group and for instructions on joining the LISTSERV email list.
This page was last updated on Tuesday, March 29, 2022
