A Sampling of Intramural Pain Research
Most of the NIH institutes and centers (ICs) are supporting or doing pain research. Following is a sampling intramural pain research being conducted at four ICs: National Center for Complementary and Integrative Health (NCCIH); National Institute of Neurological Disorders and Stroke (NINDS); National Institute on Drug Abuse (NIDA); and National Institute on Nursing Research (NINR).
NCCIH and NINDS: NCCIH Stadtman Investigator Alexander Chesler is trying to get a better understanding of the genetic and cellular mechanisms that underlie the somatosensory system, which is involved in the perception of touch, pressure, pain, and other sensations. As part of his research, he is investigating how mutations in the PIEZO2 gene affect the mechanosensation part of the somatosensory system. Unbeknown to Chesler, NINDS investigator Carsten Bönnemann, who studies the genetic basis for neuromuscular disorders in children, was also interested in the PIEZO2 gene. Bönnemann had discovered that two of his female patients with a rare neurological disorder—which caused several problems including the loss of the sensations of body position and touch—had mutations in the PIEZO2 gene. Bönnemann happened to be at a faculty meeting at which Chesler presented confocal microscopy images of Piezo2 staining in mice.
Bönnemann realized there was an opportunity for collaboration once he saw that Chesler was studying the exact same gene that was mutated in his patients (and later in other patients). As the two investigators began collaborating, Chesler extended his work from mice with Piezo2 mutations to humans.
“It’s all serendipity. I went from [being] a mouse geneticist to working with people,” said Chesler. “It changed my outlook on the role of human genomics and psychophysics,” a branch of psychology that deals with the relationships between physical stimuli and mental phenomena.
Chesler and Bönnemann have moved back and forth between patients and animal models to describe the molecular mechanisms underlying the patients’ phenotype. In addition, Chesler’s lab is now looking into the role of PIEZO2 in mechanical-pain sensation, a type of pain that is poorly understood.
Chesler and Bönnemann invited NCCIH Scientific Director Catherine Bushnell to join the collaboration to provide quantitative sensory testing and brain imaging of the patients. Bushnell’s expertise is on the brain’s role in perceiving, modifying, and managing pain. She, Chesler, and Bönnemann’s team are also studying other patients who have abnormal responses to pain. A recent patient, identified by Diana Bharucha-Goebel of NINDS and the Children’s National Health System, has them all intrigued: a teenager who can’t feel pain, but his body responds as if he does. However, his primary and secondary somatosensory cortex, the parts responsible for registering that sensation, are not activated.
“It’s a great opportunity to coordinate between clinical and basic” research, said Bushnell. “We get to translate back to animal studies and take it back to the mechanisms. The rare patients’ abnormalities help us understand normal pain processing and the mechanism of the conscious experience of pain.”

PHOTO BY: GEORGE RUDY, THINKSTOCK.COM
Catherine Bushnell (NCCIH), whose expertise is on the brain’s role in perceiving, modifying, and managing pain, has found that alternative methods of managing chronic pain such as exercise, meditation, and yoga increase tolerance to pain. Pictured: Two people practicing yoga.
Bushnell’s group is also studying alternative methods of managing chronic pain. “Lifestyle changes can alter your pain better than pharmacology,” she said. For example, rats recover faster from an injury and can tolerate pain better if they hit the “rat gym” five times a week. In addition, Bushnell’s lab has shown that meditation and yoga increase tolerance to pain. Her group also recently demonstrated that chronic pain reduces the number of opioid receptors in the brain’s pain-processing areas. This reduction in receptors may reduce the effectiveness of opioids in treating chronic pain.
Even so, opioids are often prescribed before other treatments are considered. Although the science is sound, insurance companies and medical providers haven’t caught up: Pain medications, such as opioids, are easier for doctors to prescribe and are more likely to be covered by insurance than are alternative treatments such as yoga classes.
NIDA: Amy Newman led a group that discovered antagonist compounds that target the brain’s dopamine D3 receptors (D3R), which have been implicated in reward and addiction. In animal models, the compounds reduce oxycodone self-administration, naloxone-precipitated withdrawal, and reinstatement of drug-seeking behaviors without affecting analgesia. The findings suggest that a selective D3R blockade may reduce the development of opioid dependence without diminishing the effectiveness of prescription pain killers. In addition, the lead molecules identified may offer a potential treatment for opioid-use disorders. This NIH technology has been licensed by pharmaceutical-industry partners to further develop these promising molecules for the prevention and treatment of opioid misuse and addiction.
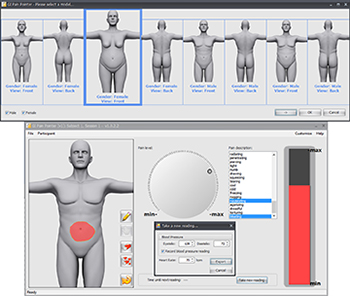
Wendy Henderson’s group (NINR) developed the Gastrointestinal Pain Pointer (GIPP), a new computerized tool for assessing the location and severity of abdominal pain. The Gastrointestinal Pain Pointer, developed by W.A. Henderson and A.P. Zuccolotto, Public Health Service Invention #E-175-2010; licensing available with Psychology Software Tools, Inc. (Image from: M.M. Heitkemper and W.A. Henderson, “Gastrointestinal Symptom Science and Assessment,” Using Nursing Research to Shape Health Policy, Editors P.A. Grady and A.S. Hinshaw, Springer Publishing Co., LLC, New York, 2017)
NINR: Because pain is a difficult condition to characterize, finding a standardized way to classify pain intensity is a clinical goal outlined by the HEAL initiative. Wendy Henderson’s group, in NINR’s Biobehavioral Branch, developed the Gastrointestinal Pain Pointer (GIPP), a new computerized tool for assessing the location and severity of abdominal pain. The tool combines subjective scores with objective measures such as heartrate and can be used to detect subclinical gastrointestinal issues, such as inflammation, which often underlies chronic pain. This scoring method can be translated to clinical facilities and provide benefits to patients and researchers studying chronic GI pain.
Wish list. Funding an intramural NIH center for pain is at the top of the wish list for Bushnell, Chesler, and others. Chesler emphasized the benefits of having an effective, centralized place where resources and expertise could be shared.
“Pain has been an orphan for decades,” said Bushnell. “It wasn’t a disease. It didn’t kill you. But now with the [opioid] epidemic it turns out that it does.”
There are a number of ways to find other pain researchers at NIH.
Scientific Interest Groups: See write-up for the PAIN Scientific Interest Group in this issue of the NIH Catalyst. For a list of all SIGs, go to https://oir.nih.gov/sigs.
The Intramural Research Program (IRP) Website: The IRP website includes individual pages for all of the IRP’s approximately 1,050 principal investigators, complete with contact information, descriptions of their research, and lists of selected publications. You can search for “pain” on the principal investigators page or on the main IRP page.
The NIH Intramural Database (NIDB): The NIDB contains a wide array of information about the activities of NIH researchers, including annual reports detailing their current projects and related publications. Simply selecting a year and an IC (or “All ICs”) and entering a search term yields a list of related researchers and their projects.
NOTE: STOCK IMAGE USED AS THUMBNAIL IS BY ROMOLO TAVANI, THINKSTOCK.COM
This page was last updated on Wednesday, April 6, 2022
