Research Briefs
NCI: CELLULAR IMMUNOTHERAPY TARGETS A COMMON HUMAN CANCER MUTATION

CREDIT: NCI
A color illustration of the KRAS protein, with blue helices wrapping around a core of balls and sticks.
In a study of an immune therapy for colorectal cancer that involved a single patient, a team of researchers at NCI identified a method for targeting the cancer-causing protein produced by a mutant form of the KRAS gene. This targeted immunotherapy led to cancer regression in the patient. Mutations in the KRAS gene are thought to drive 95 percent of all pancreatic cancers and 45 percent of all colorectal cancers. The team isolated tumor infiltrating lymphocytes (TILs) that targeted the KRAS G12D mutation from tumor nodules in the patient’s lungs that developed after colorectal cancer cells had spread to the lungs.
The isolated TILs were grown in the laboratory to large numbers and then infused into the patient intravenously. After the TIL infusion, all seven metastatic lung nodules in the patient regressed, and the regression persisted for nine months. After nine months, one of the lesions progressed and was surgically removed. This lesion was found to have lost a segment of chromosome 6 that includes a gene known as HLA-C*0802, which is involved in antigen presentation. If the immune system recognizes the antigen as abnormal or foreign, an immune response against it will be mounted. In this case, because of the loss of the segment of chromosome 6, the immune system was unable to recognize the cancer cells as being abnormal, and they were able to escape immune attack and continue to thrive. Since the lesion was removed, however, the patient has been disease-free for over eight months. The authors of the study note that this work is a proof-of-principle and that the next step is to see whether the method proves effective in other patients. (NIH authors: E. Tran, P.F. Robbins, Y. Lu, T.D. Prickett, J.J. Gartner, L. Jia, A. Pasetto, Z. Zheng, S. Ray, E.M. Groh, I.R. Kriley, and S.A. Rosenberg, N Engl J Med 375:2255–2262, 2016)
NIDCD: HEARING-LOSS PREVALENCE DECLINING IN U.S. ADULTS
Hearing loss among U.S. adults aged 20 to 69 has declined over the past decade, even as the number of older Americans continues to grow, according to a new NIH study. These findings also confirm that hearing loss is strongly associated with age and other demographic factors such as sex, race and ethnicity, and education. Researchers at NIDCD compared hearing health data collected as part of the National Health and Nutrition Examination Survey (NHANES) over two time periods: 2011–2012 and 1999–2004. The researchers do not know why hearing-loss prevalence is declining but speculate possible factors include fewer manufacturing jobs, increased use of hearing protectors, less smoking, and advances in health including better medical care to manage risk factors associated with hearing loss. (NIH authors: H. Hoffman, K. Losonczy, and C. Themann, JAMA Otolaryngol Head Neck Surg DOI:10.1001/jamaoto.2016.3527)
NCCIH: VETERANS ENDURE HIGHER PAIN SEVERITY THAN NONVETERANS
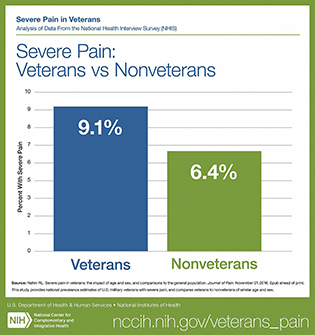
CREDIT: NCCIH
Veterans reported more severe pain than nonveterans.
American veterans experience higher prevalence of pain and more severe pain than nonveterans, with young and middle-aged veterans suffering the most, according to a new analysis of the National Health Interview Survey (NHIS) by the lead epidemiologist at the NCCIH. This survey provides the first national estimate of severe pain associated with painful health conditions in veterans and nonveterans and underscores the importance of sustaining efforts to monitor and manage pain among veterans. The analysis showed that veterans were about 40 percent more likely to experience severe pain than nonveterans, and younger veterans were substantially more likely to report suffering from severe pain than nonveterans. The analysis is based on data from the 2010–2014 NHIS, in which 67,696 adults (6,647 veterans and 61,049 nonveterans) responded to questions about the persistence and intensity of self-reported pain during the three months prior to the survey. Most veteran participants were men (92.5 percent), whereas the majority of nonveteran participants were women (56.5 percent). More research is needed to generate additional evidence-based options for veterans managing pain. Over time this research may help nonveterans as well. (NICCH author: RL Nahin, J Pain DOI:10.1016/j.jpain.2016.10.021)
NINDS AND NIAID: CEREBRAL MALARIA’S DEADLY AGENTS
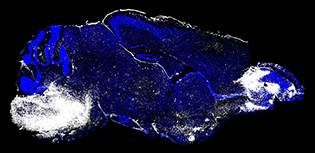
IMAGE COURTESY OF DORIAN MCGAVERN (NINDS) AND PHILLIP SWANSON II (NIAID)
This image shows the brain of a mouse with cerebral malaria. White regions (left, brainstem and right, olfactory bulb) indicate areas of neuronal cell death and vascular leakage.
Using state-of-the-art brain-imaging technology, NINDS and NIAID scientists filmed what happens in the brains of mice that developed cerebral malaria (CM). The results reveal the processes that lead to fatal outcomes of the disease and suggest an antibody therapy that may treat it.
To see inside the brain of a mouse infected with the parasite that causes CM, the researchers used intravital microscopy, which allowed them to watch cells in action. To determine which parts of the brain were affected by these events, the researchers injected mice with dyes that marked dead cells and leaks in blood vessel. The results indicated that the brain regions with the most damaged vessels and cell death were the olfactory bulb (the area involved in sensing smell) and crucially, the brainstem, an area that controls such vital functions as breathing and heart rate.
In another set of experiments, the group tested a potential therapy to see whether it could be used to remove the CD8+ T cells from vessel walls. Initially, the CD8+ T cells began to interact with the cerebral blood vessels in the CM mice. Then the scientists treated the mice with two FDA-approved intravenous drugs that block the molecules that CD8+ T cells use to attach to blood vessels. Within 30 minutes of the treatment, the CD8+ T cells broke off from the blood vessels and could not stick to them, preventing the fatal brain swelling in all of the treated mice. These findings suggest that the interactions between CD8+ T cells and blood vessels lead to death from CM and preventing that binding may increase survival from the disease.
In future studies, the researchers will examine how the interaction between CD8+T cells and cerebral vessels causes blood leakage and ways in which the brain recovers from CM infection. In addition, the live-action imaging technology from this study may be used to watch ways in which other mosquito-borne illnesses, such as those caused by the Zika and dengue viruses, affect the brain. (NIH authors: P.A. Swanson, G.T. Hart, M.V. Russo, T. Yazew, M. Peña, S.K. Pierce, and D.B. McGavern, PLoS Pathog DOI:10.1371/journal.ppat.1006022)
NCATS, CC, AND NIAID: NEW RAPID-SCREENING TEST MAY IDENTIFY POTENTIAL THERAPIES AGAINST DRUG-RESISTANT BACTERIA
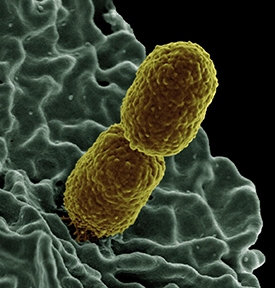
CREDIT: NIAID
Colorized scanning-electron micrograph showing carbapenem-resistant Klebsiella pneumoniae interacting with a human white blood cell.
Researchers at the NCATS, the NIAID, and the Clinical Center have created a new way to identify drugs and drug combinations that may potentially be useful in combating infections that are resistant to many different antibiotics. They developed an assay to rapidly screen thousands of drugs to determine how effective they were against a variety of types of resistant bacteria. The screening method provides a potential new approach to repurpose known drugs and compounds to help deal with powerful, hospital-borne infections as well as with emerging infectious diseases. Of the approximately 4,000 approved drugs and other biologically active compounds that were screened, 25 suppressed the growth of two drug-resistant strains of Klebsiella pneumoniae that have become resistant to most major types of antibiotics. Drug-resistant Klebsiella has been a source of fatal infections in many hospitals across the country.
The researchers also used the screening test to gauge the effectiveness of combinations of drugs against antibiotic-resistant bacteria in this study. They found three different three-drug combinations that were effective against 10 common strains of multi-drug-resistant bacteria. In the future, this assay may provide crucial information to mount a rapid response to emerging infectious diseases. (NIH authors: W. Sun, R.A. Weingarten, M. Xu, N. Southall, S.Dai, P.Shinn, P.E. Sanderson, P.R. Williamson, K.M. Frank, and W. Zheng, Emerg Microbes Infect 5:e116, 2016; DOI:10.1038/emi.2016.123)
NIAID: TESTING OF INVESTIGATIONAL INACTIVATED ZIKA VACCINE IN HUMANS BEGINS
The first of five early-stage clinical trials to test the safety and ability of an investigational Zika vaccine candidate called the Zika purified inactivated virus (ZPIV) vaccine to generate an immune system response has begun at the Walter Reed Army Institute of Research (WRAIR) Clinical Trial Center (Silver Spring, Maryland). Scientists with WRAIR, part of the U.S. Department of Defense, developed the vaccine. NIAID is co-funding the phase 1 clinical trial with WRAIR, serving as the regulatory sponsor and providing other support. WRAIR, NIAID, and the Biomedical Advanced Research and Development Authority, part of the HHS Office of the Assistant Secretary for Preparedness and Response, have established a joint Research Collaboration Agreement to support the development of this vaccine.
NIAID’s Vaccine Research Center (VRC) will test the ZPIV vaccine candidate as a booster vaccination to its DNA Zika vaccine candidate, which entered phase 1 clinical trials in August. The next part of the study, which will enroll 60 additional participants ages 18–50 years, will take place at the NIH Clinical Center, the Center for Vaccine Development at the University of Maryland School of Medicine’s Institute for Global Health (Baltimore), and Emory University (Atlanta). Julie Ledgerwood, chief of the VRC’s clinical-trials program, will serve as principal investigator. To read more go to https://www.nih.gov/news-events/news-releases/testing-investigational-inactivated-zika-vaccine-humans-begins.
NHLBI: NEW METHOD FOR PERFORMING AORTIC-VALVE REPLACEMENT PROVES SUCCESSFUL IN HIGH-RISK PATIENTS
Scientists at NHLBI have developed a new, less-invasive way to perform transcatheter aortic-valve replacement (TAVR), a procedure widely used to treat aortic-valve stenosis, a lethal heart condition. The new approach, called transcaval access, will make TAVR more available to high-risk patients, especially women, whose femoral arteries are too small or too diseased to withstand the standard procedure. Transcaval access, which can be performed in awake patients, involves electrifying a small wire so that it crosses between neighboring blood vessels in the abdomen. The technique calls for making large holes in both the abdominal aorta and the inferior vena cava, which physicians previously considered dangerous because of the risk of fatal bleeding. The new method was tested in a trial on 100 patients at 20 hospitals across the United States. Researchers said it proved successful in 99 patients. (NIH author: R.J. Lederman, J Am Coll Cardiol 63:2795–2804, 2016)
NHGRI, NEI: ACCLERATING WOUND HEALING
NHGRI and NEI researchers have identified a novel role for a protein known as heat shock protein 60 (Hsp60), finding that it is critical in tissue regeneration and wound healing. The study found that topical treatment of an Hsp60-containing gel dramatically accelerates wound closure in a diabetic mouse model. These results may help in the development of effective therapeutics for accelerating wound closure in diabetic patients as well as for normal wound healing and scar reduction. When Hsp60 was injected directly into the site of injury, the tissue surrounding the wound started to regenerate faster.
The most striking finding was that applying a topical treatment of the Hsp60 protein to a puncture wound in diabetic mice stimulated nearly complete healing after only 21 days. Mice without the treatment showed little or no improvement during the same time. Although promising, this finding has only been shown in mice and is yet to be tested in humans. (NIH authors: W. Pei, S.C Huang, L. Xu, B. Liu, J. Sinclair, J. Idol, G.K. Varshney, R.B. Nussenblatt, and S.M. Burgess, NPJ Regen Med DOI:10.1038/npjregenmed.2016.13)
NHGRI, NICHD: GENE THERAPY SHOWS PROMISE FOR TREATING NIEMANN-PICK DISEASE TYPE C1
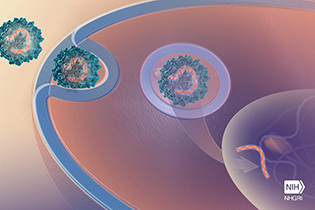
CREDIT: NHGRI
The gene therapy involved inserting a functional copy of the NPC1 gene into mice with the disease. The treated animals were then found to have less-severe Niemann-Pick symptoms.
For the first time, NIH researchers at NHGRI and NICHD have demonstrated in mice that gene therapy may be the best method for correcting the single faulty gene that causes Niemann-Pick disease, type C1 (NPC1). They inserted a functional copy of the NPC1 gene into mice with the disease; the treated animals were then found to have less-severe NPC1 symptoms.
Niemann-Pick disease is a rare and fatal disorder of the central nervous system (the brain and spinal cord) that has no cure. The disease occurs when a faulty housekeeping gene fails to remove cell waste, such as lipids and cholesterol. The accumulation of waste in the spleen, liver, and brain causes progressive deterioration in intellectual and motor functions. It also shortens patients’ lives; people with Niemann-Pick disease typically die in their teens. There are several types of Niemann-Pick disease; this study focused on mice that had been bred with a faulty NPC1 gene to model NPC1.
The researchers’ goal was to correct the faulty NPC1 gene in as many cells and organs as possible, with a strong focus on the brain. To do this, they used a non-disease-causing virus called the adeno-associated virus serotype 9 (AAV9) to transfer functioning NPC1 to the cells. AAV9 with a functioning NPC1 gene successfully crossed the blood–brain barrier, reaching cells in the brain and elsewhere. Once inside cells, the normal NPC1 gene was then able to make the functional NPC1 protein to correct the cell defects. With a single injection, mice improved motor coordination, gained weight, and lived longer compared with those that did not have this gene therapy. (NIH authors: R.J. Chandler, I.M. Williams, A.L. Gibson, A.A. Incao, B.T. Hubbard, F.D. Porter, W.J. Pavan, and C.P. Venditti; Hum Mol Genet DOI:10.1093/hmg/ddw367)
NCI AND FDA: NO SAFE LEVEL OF SMOKING
People who consistently smoked an average of fewer than one cigarette per day over their lifetimes had a 64 percent higher risk of earlier death than never smokers, and those who smoked between one and 10 cigarettes a day had an 87 percent higher risk of earlier death than never smokers, according to a new NCI study. Risks were lower among former low-intensity smokers compared with those who still smoked, and risk fell with earlier age at quitting. When researchers looked at specific causes of death among study participants, a particularly strong association was observed for lung-cancer mortality. Those who consistently averaged fewer than one cigarette per day over their lifetimes had nine times the risk of dying from lung cancer as never smokers. Among people who smoked between one and 10 cigarettes per day, the risk of dying from lung cancer was nearly 12 times that of never smokers.
The scientists analyzed data on over 290,000 adults in the NIH-AARP Diet and Health Study. Low-intensity smoking was defined as 10 or fewer cigarettes per day. All participants were age 59 to age 82 at the start of the study. The researchers pointed out that the study had several limitations: It relied on people recalling their smoking history over many decades, which introduced a degree of uncertainty into the findings; the number of consistent low-intensity smokers was relatively small; participants were mostly white and in their 60s and 70s, so the smoking patterns collected in the study reflect only a particular set of demographics; and the study also lacked detailed information about usage patterns among participants who reported smoking less than one cigarette per day. The findings, however, indicated that smoking even a small number of cigarettes per day has substantial negative health effects and provide further evidence that smoking cessation benefits all smokers, regardless of how few cigarettes they smoke. (NIH authors: M. Inoue-Choi, L. Liao, P. Hartge, N. Caporaso, and N. Freedman; FDA author: C. Reyes-Guzman, JAMA Intern Med DOI:10.1001/jamainternmed.2016.7511)
NICHD: CIGARETTE SMOKING DURING PREGNANCY LINKED TO CHANGES IN BABY’S IMMUNE SYSTEM
Infants whose mothers smoked cigarettes throughout pregnancy had higher levels of the immune-system protein interleukin-8 (IL-8) at birth, according to a study led by researchers at the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD). IL-8 is a protein that is known to promote inflammation. The researchers evaluated information from 2,903 mothers who consented to the additional analysis of their child’s residual dried blood sample, which had been collected for newborn screening tests two to three days after birth. About 12 percent of the women had smoked during pregnancy, and some quit smoking during their first trimester. However, 43 percent of the smokers continued to smoke throughout pregnancy.
The study is among the largest to show that smoking throughout pregnancy leads to changes in an infant’s immune system, even at birth. Future studies, which the researchers are planning, will need to determine if a newborn’s levels of IL-8 can predict future smoking-related health conditions such as asthma. Being able to identify children at risk for these conditions would allow for early monitoring, possible prevention, and early treatment. [NIH authors: N. Chahal A. Ghassabian, K.A. Michels, E.H. Yeung; Nicotine and Tobacco Research DOI: 10.1093/ntr/ntw324 (2016)]
This page was last updated on Saturday, January 18, 2025
