Research Briefs
NHGRI, NHLBI, NIAID, NIAMS, NCI, NIDDK, CC: DISCOVERY OF A GENETIC DISORDER THAT CAUSES STROKES AND VASCULAR INFLAMMATION IN CHILDREN
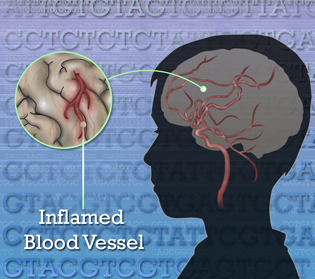
NIH researchers have identified gene variants that cause a rare syndrome of sporadic fevers, skin rashes, and recurring strokes, beginning early in childhood. The team’s discovery coincides with findings by an Israeli research group that identified an overlapping set of variants of the same gene in patients with a similar type of blood-vessel inflammation.
Jonathan Bailey, National Human Genome Research Institute, NIH
A variation in the gene that codes for a key blood-vessel enzyme causes a rare disease in children that makes them prone to fevers, rash, and strokes. The discovery of what underlies this blood-vessel disease may also benefit efforts to treat and prevent stroke in general.
The NIH group first encountered a patient with the syndrome approximately 10 years ago. The patient, then 3 years old, experienced fevers, skin rash, and strokes that left her severely disabled. Because there was no history of a similar illness in the family, the NIH group did not at first suspect a genetic cause, and it treated the patient with immunosuppressive medications.
Several years passed, but then two unrelated children with very similar symptoms came to NIH. The researchers began to suspect a common genetic cause and embarked on a medical odyssey that has led not only to a diagnosis, but also to fundamental new insights into blood-vessel disease. Intramural scientists from seven NIH Institutes and Centers teamed up to sequence and analyze the exomes—the protein-coding part of the genomes—of all three affected children and their unaffected parents. That was no small feat, because although the exome makes up less than 2 percent of the human genome, it still contains about 30 million DNA letters. Sequencing efforts of this scale have only become feasible in the last few years, thanks to the development of faster, cheaper sequencing technologies.
When the researchers examined the genetic data, they discovered all three children had two mutated copies of the CECR1 gene—one copy from each of their parents. In contrast, their parents each carried one normal copy and one mutated copy of the CECR1 gene.
The CECR1 gene codes for an enzyme called adenosine deaminase 2 (ADA2), which is crucial for blood-vessel development and maintaining the balance of key immune cells called monocytes and macrophages. The mutated copies of CECR1 found in the young patients impair their ability to produce the ADA2 enzyme. This ADA2 deficiency, the researchers found, leads to vascular and immune system abnormalities that promote a vicious cycle of inflammation that, in turn, raises the risk of stroke.
The discovery of what underlies this rare blood-vessel disease, which researchers have dubbed “Deficiency of ADA2,” or DADA2, may also benefit efforts to treat and prevent a much more common vascular disorder: stroke. Intriguingly, at least one previous study has found that adults who carry one CERC1 mutation may face an increased risk of late-onset stroke. Building on the new findings, it is now possible to delve further into the role of the ADA2 pathway in stroke and other blood-vessel disorders. (NIH authors: Q. Zhou, D. Yang, D.L. Kastner, M. Boehm, I. Aksentijevich, et al.; N Eng J Med DOI:10.1056/NEJMoa1307361)
NIDDK, NICHD, NCI: SHIVERING MAY BOOST HEAT-PRODUCING BROWN FAT IN HUMANS
When NIH researchers explored how hormones regulate physiologic responses to cold exposure in humans, they uncovered an intriguing relationship between shivering, brown fat, and exercise. Mammals maintain body temperature through biological processes that reduce heat loss and increase heat production during cold exposure. Brown fat and muscle are central to thermal homeostasis. In contrast to ordinary white fat, whose primary function is to store excess energy as lipids, brown fat is a unique heat-producing tissue, specializing in burning its stored lipids and releasing energy as heat. On the other hand, muscle generates heat during exercise or shivering. How brown fat and muscle communicate to maintain body temperature has been poorly understood.
In this study, healthy volunteers were exposed to cold temperatures until shivering occurred. Blood samples were collected during cooling to measure concentrations of different hormones. These values were then compared with those obtained during an exercise test.
The researchers found the hormones irisin and fibroblast growth factor 21 (FGF21) to be cold-stimulated, the former from shivering muscle and the latter from activated brown fat. Exercise and shivering increased irisin concentrations to similar extent. The team then treated human fat cells with these hormones in the laboratory and found that irisin and FGF21 both transformed human white fat cells into heat-producing brown fat cells.
Since the discovery of irisin in 2012 (in an NIH-funded study), scientists have puzzled over why exercise, an energy-expending process, would stimulate a hormone that produces heat. The current study suggests exercise could be mimicking shivering through muscle contraction, but the evolutionary origin of irisin could be a cold-activated endocrine system that turns white fat into heat-producing brown fat to protect humans from cold temperatures. From a clinical point of view, the cold-activated hormones irisin and FGF21 hold promise as potential treatment targets for obesity and related metabolic disorders. (NIH authors: P. Lee, J. Linderman, S. Smith, R. J. Brychta, J. Wang, C. Idelson, R.M. Perron, C.D. Werner, G.Q. Phan, U.S. Kammula, E. Kebebew, K. Pacak, K.Y. Chen, F.S. Celi; Cell Metab 19:302–309, 2014)
NIDCR: P38 IS HIGHLY ACTIVE IN HEAD AND NECK CANCERS
Better treatment options are needed for people diagnosed with head and neck cancers because the current therapies of surgery, radiation therapy, and chemotherapy control cancer at the cost of normal tissues, sometimes damaging them permanently. An NIDCR-led team of scientists reported that p38 kinase is active in head and neck cancer cells and blocking p38 may help prevent cancers from growing. Although p38 is known to play a role in breast and bladder cancers, these results may be the first to show that p38 also plays a highly active role in head and neck cancers.
The team tested SB203580, a drug known to block p38 activity. As expected, SB203580 reduced the growth of head and neck cancer cells in the lab. When the NIDCR team used the drug SB203580 to treat human head and neck cancers that had been transplanted into lab mice, SB203580 shrank the cancers.
The next step will be to test a new generation of drugs that inhibit p38. The team has developed new mouse models of metastatic oral cancer and hopes to demonstrate that preventing the activation of p38 will not only diminish the growth of the oral cancer and the new blood vessels that feed the cancer, but will also reduce the cancer’s ability to spread to nearby lymph nodes and spread throughout the body. (NIH authors: K. Leelahavanichkul, P. Amornphimoltham, A.A. Molinolo, J. S. Gutkind; Mol Oncol 8:105–118, 2014)
NIDCR: CELLS THAT BREAK DOWN COLLAGEN
NIDCR researchers collaborated with an international team of scientists to visualize collagen protein within living tissues and determine which cells degrade it. They were surprised to find that the most important cell type in animals (mice) was different from the most common cell type identified in previous in vitro laboratory studies.
Although many types of cells can make the collagen-degrading enzymes known as collagenases, the scientists reported that they identified the most important cell type as a type of blood cell known as an M2-like macrophage because it shares characteristics with M2 macrophages. Although one of the usual functions of an M2 macrophage is to clean up cell debris by engulfing and degrading it, in the earlier in vitro studies, fibroblasts—not macrophages—were the cell type that had been most important for collagen internalization and degradation. Their research thus supports the hypothesis that M2-like macrophages are involved in tissue repair.
The researchers next want to determine whether the extensive tissue-remodeling process that occurs around tumors and the accompanying collagen degradation is principally caused by cancer cells, tumor-associated fibroblasts, or macrophages. Previous studies have suggested that tumor-associated M2-like macrophages may promote the growth of cancer and its invasion of nearby tissues. Elucidating the mechanism of collagen degradation in cancer is the first step in devising therapies that could prevent invasion of cancer. (NIDCR authors: D.H. Madsen, D. Leonard, A. Masedunskas, A. Moyer, H.J. Jürgensen, D.E. Peters, P. Amornphimoltham, A. Selvaraj, R. Weigert, T.H. Bugge; J Cell Biol 202:951–966, 2013)
OTT: NIH’S SUCCESS IN DRUG DEVELOPMENT
Compared with other U.S. public-sector research institutions, the NIH Intramural Program (IRP) has contributed inventions that have had a disproportionately greater impact on the overall number of products produced (particularly vaccines, cancer therapeutics and in vivo diagnostics), drugs granted orphan status, and drugs granted priority review because they offer major advances in treatment.
The drugs referred to are powerhouse medications and vaccines such as paclitaxel (Taxol), live oral tetravalent Rotavirus vaccine (RotaShield, now being tested in Africa), bortezomib (Velcade), quadrivalent human papillomavirus vaccine (Gardasil), and darunavir (Prezista). The total global net sales of drugs using inventions developed by the NIH-IRP was nearly $7 billion in 2010. In the past two decades, the NIH-IRP has contributed more than 14 percent of the total number of drugs brought to the market under commercial licenses from public-sector research institutions while receiving about 11 percent of all NIH research funds to these institutions (not including the non-NIH funding universities receive). (OTT authors: S.K. Chatterjee, M.L. Rohrbaugh; Nat Biotechnol 32:52–58, 2014)
This page was last updated on Wednesday, April 27, 2022
