The Festival that Almost Wasn’t
Report from the 2013 Research Festival

DARRYL LEJA, NHGRI
This design represents the two 60th anniversary celebrations that the 2013 Research Festival recognized: that of the NIH Clinical Center, which opened in July 1953; and of James Watson and Francis Crick’s landmark paper that first described the DNA double-helix structure, published in Nature on April 25, 1953.
Neither snow, nor rain, nor the gloom of a government shutdown could keep NIH’s Research Festival from happening. It was just postponed a little.
Led by co-chairs Dan Kastner, the scientific director of the National Human Genome Research Institute (NHGRI), and Luigi Ferrucci, the scientific director of the National Institute on Aging (NIA), the festival served up a feast of basic science and translational medicine.
Although the festival was originally scheduled for early October, the 16-day government shutdown forced the organizers to rapidly reschedule the events for November 6 to 8 instead. Most of the planned events took place—including the plenary session, many concurrent symposia, and a much-anticipated Scientific Directors Poster Session and Cook-off—but the Vendor Tent Show had to be rescheduled for the spring, and the National Graduate Student Research Conference had to be cancelled.
“The silver lining, perhaps, of the shutdown was that NIH emerged as one of a very short list of government activities that everybody agreed has incredible value,” said NIH Director Francis Collins in his opening remarks at the plenary session.
This year’s festival celebrated two 60th anniversaries: that of James Watson and Francis Crick’s landmark paper that first described the DNA double-helix structure, published in Nature on April 25, 1953 (Nature 171:737–738, 1953); and of the NIH Clinical Center, which was dedicated on July 2, 1953, and admitted its first patients four days later on July 6. In fact, the whole Research Festival took place in the Clinical Center—Masur Auditorium and in the newly opened Foundation for Advanced Education in the Sciences (FAES) Academic Center—instead of the Natcher Building (Building 45), where it’s been held for years.
“We really wanted to make a translational connection,” said Ferrucci. “We wanted to invite the people who are in the labs—our community—to celebrate instances where we can translate [basic science] into human health.”
The plenary session featured three talks that highlighted how the focuses of the Festival—modern translational science at the Clinical Center and the study of DNA—have become intertwined in 21st century research. The first presentation, by Kastner, illustrated how new technologies have allowed researchers to discover the genetic basis of rare diseases using smaller and smaller cohorts of patients.
“With the advent of whole-exome sequencing and whole-genome sequencing, we have tools [that] allow us to take these cohorts of relatively rare patients, or even small families, and crack those cases in ways we weren’t able to before,” said Kastner. One of the cases he highlighted involved a young child who came to the NIH Clinical Center with early-onset stroke and fever. While the child was treated with steroids and began to get better, it wasn’t until years later, when another child with the same symptoms came to the Clinical Center, that a sequencing analysis on just six individuals revealed that a single gene, with recessive inheritance, was responsible for the disorder. Mutations in the same gene, encoding the adenosine deaminase protein ADA2, were later found in several more patients with similar symptoms. “I think there’s a very bright future in being able to solve some of those medical mysteries,” said Kastner. “It allows us to . . . begin to parlay that [knowledge] into targeted therapies for those patients.”
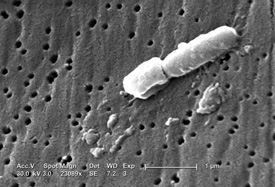
NHGRI senior investigator Julie Segre described another medical mystery that she and other NIH intramural investigators helped solve: What was causing the antibiotic-resistant Klebsiella pneumoniae infections that occurred at the NIH Clinical Center during 2011? In June of that year, a patient colonized with the bacterium was transferred to the Clinical Center; several weeks after her discharge, other patients at the Clinical Center were found carrying the same strain of antibiotic-resistant bacteria.
Traditional techniques for identifying bacteria can only broadly identify the strain. Segre, however, used new techniques for DNA sequencing to sequence the entire genome of each patient’s infection, and she detected single base-pair changes. By tracing the path of infection through the hospital from patient to patient, she could help identify which additional methods were needed to eliminate the bacteria from the Clinical Center. “What has really driven me into this area of research is the extent to which health-care-associated infections contribute to the public’s concern about seeking health care,” said Segre. “Hospital infection control, with mandatory testing, is going to be crucial.” (To read more, see the November-December 2012 issue of the NIH Catalyst.)
JANICE CARR, CDC
Julie Segre (NHGRI) described her team’s efforts to determine the cause of antibiotic-resistant Klebsiella pneumoniae infections that were occurring among gravely ill, immune-compromised patients in the NIH Clinical Center in 2011. This scanning-electron micrograph reveals some of the ultrastructural morphologic features of the bacterium.
Next it was Cynthia Dunbar’s turn to describe her own detective work. Dunbar, a senior investigator in the National Heart, Lung, and Blood Institute (NHLBI), studies how hematopoietic stem cells produce cells that repopulate the immune system. Her model is the rhesus macaque because its hematopoietic and immune systems closely match that of humans. After introducing genetically “barcoded” hematopoietic stem cells into the macaques, she applied high-throughput sequencing techniques to detect cells derived from the original stem cells. She found that myeloid cells and B cells are closely related in the hematopoietic hierarchy, but there is no evidence for a common lymphoid progenitor. Most interesting were her findings regarding natural killer cells, which showed an ontogeny completely distinct from T, B, and myeloid cells at least through six months post-transplantation. “The molecular techniques [allow] us to approach the body like science fiction investigators in ways we never imagined would be possible,” said Dunbar.
After the plenary session, the Research Festival continued with poster sessions and concurrent symposia and, of course, the Scientific Directors Poster Session and Cook-off, a competition for the best poster presentation and the best home cooking.
Ferrucci “was the one who suggested the cook-off,” said Kastner. “He [said he] would prepare a batch of traditional Italian biscotti and he threw down the gauntlet to the other scientific directors, to see [whether] they could measure up to his standards of culinary excellence.” The prize for both best poster and best cooking went to Kastner—the latter for home-baked chocolate chip cookies.
In the end, the 2013 NIH Research Festival weathered the bumps of the government shutdown and subsequent rescheduling.
“It was terrific to have an opportunity for people to get together again after the shutdown, to make scientific and other connections amongst the community at the NIH,” said Kastner.
“I thought [the festival] was re-energizing,” said Ferrucci. “I felt that spirit come out . . . the enthusiasm, of just doing the science [and] finding and showing your results.”
- To see a video of the plenary session, go to http://videocast.nih.gov/launch.asp?18152
- To watch a YouTube video of the Research Festival highlights, go to http://youtu.be/TilycWcU_zk

ERNIE BRANSON
Cutting the ribbon to celebrate the opening of FAES’s new space in Building 10, where the Research Festival took place: (from left to right) FAES Executive Director Christina Farias; CC Director John Gallin; NIH Director Francis Collins; Deputy Director for Intramural Research Michael Gottesman; and Angela Gronenborn, president of FAES (former senior investigator in NIDDK and now at the University of Pittsburgh)

ERNIE BRANSON
In this year’s Research Festival, even the Scientific Directors (SDs) presented posters. Dan Kastner, Research Festival co-chair and NHGRI SD, won first place for his poster and for his chocolate chip cookies.

ERNIE BRANSON
Luigi Ferrucci, fellow co-chair and NIA SD (right) explained his poster to Deputy Director for Intramural Research Michael Gottesman.
Posters and Cookies and SDs, Oh My
For the first time in the history of the NIH Research Festival, scientific directors (SDs) presented scientific posters and competed in a cooking competition. The directors welcomed the change of pace from their administrative duties and enjoyed lively discussions with fellows and trainees about their true passion–the research in their labs.

ERNIE BRANSON
NICHD Scientific Director Constantine Stratakis (left) explained his poster to NHGRI Deputy Scientific Director Paul Liu. Stratakis’s poster provided an overview of recent advances in understanding Cushing syndrome.
Constantine Stratakis (NICHD) presented a comprehensive overview of recent advances in understanding Cushing syndrome, a disorder characterized by obesity and muscular weakness due to high concentrations of the hormone cortisol. His work represented the combined efforts of more than 100 trainees in his lab over the past 20 years.
One of the more recent recruits to NIH, NCCAM’s scientific director Catherine Bushnell, presented a poster that highlighted changes that occur in the brains of patients who suffer from chronic pain. Even Deputy Director for Intramural Research Michael Gottesman joined in the fun (his lab is in NCI)—his poster featured the analysis of biomarkers that determine drug resistance in cancer cells.
For many of the fellows who attended the session, it was a rare occasion to turn the tables on the SDs and quiz them on their scientific knowledge. And, of course, judges evaluated the posters and voted by secret ballot to determine the winner. To ensure fairness, each of the institutes nominated a fellow to serve on the panel of judges.
One judge viewed the poster session not only as a look into past work by the SDs but also as a peek into the future of the research direction of the NIH. “This is a great opportunity to pose scientific questions to the SDs,” said Elyssa Monzack (NIDCD). “These are people who have a say in what is important in their fields.”
Participants and judges were also invited to savor a spectacular variety of homemade treats. Much of the spread was inspired by the directors’ cultural heritages, reflecting the vibrancy and international diversity found in labs throughout NIH. In addition to many variations of the traditional American chocolate-chip cookie, attendees were treated to a cornucopia that included rugelach, Greek baklava, biscotti, Hungarian sausage, Louisiana pecan kisses, and chocolate-covered strawberries.
The judges’ work was cut out for them, into bite-sized pieces, with the culinary creations also being an item of contention. “Judging desserts and science are not the same,” said Monzack. “But [they] are equally . . . delightful.”
And the winners?
Well, there was only one winner. Congratulations, and bragging rights among the directors, goes to Daniel Kastner, Research Festival co-chair and scientific director of NHGRI. Kastner took the prize with his poster describing his group’s recent identification of a new autoinflammatory disease, adenosine deaminase 2 deficiency, in which the deficiency of a protein leads to a spectrum of vascular disease and systemic inflammation. And he won over both the crowd and the judges with his “Butterlicious Chocolate Chip Crepes with Funfetti Frosting.”
As would be expected for any dedicated scientist, Kastner thoroughly experimented and optimized his methods in the kitchen. For a detailed analysis of how Kastner engineered his award-winning desserts, check out the video at http://youtu.be/hjuo4D_WDdw.
This page was last updated on Wednesday, April 27, 2022
