NEI: Using Artificial Intelligence for Quality Control of Stem Cell–Derived Tissues
Technique key to scaling up manufacture of therapies from induced pluripotent stem cells
Researchers from the National Eye Institute (NEI) and the National Institute of Standards and Technology (NIST) have used artificial intelligence (AI) to evaluate stem cell–derived “patches” of retinal pigment epithelium (RPE) tissue for implanting into the eyes of patients with age-related macular degeneration (AMD), a leading cause of blindness.
The proof-of-principle study helps pave the way for AI-based quality control of therapeutic cells and tissues. The method was developed by researchers at NEI and NIST and is described in a report published online November 12, 2019, in the Journal of Clinical Investigation.
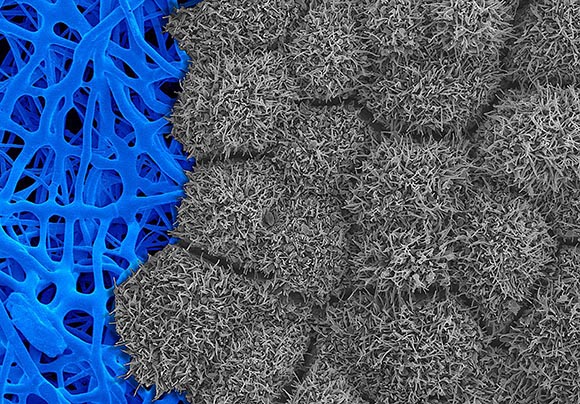
CREDIT: NATHAN HOTALING, NCATS
Scanning electron micrograph showing induced pluripotent stem cell–derived retinal pigment epithelium tissue (gray) cultured on a fiber-based scaffold (blue).
“This AI-based method of validating stem cell–derived tissues is a significant improvement over conventional assays, which are low-yield [and] expensive and require a trained user,” said Kapil Bharti, a senior investigator in NEI’s Ocular and Stem Cell Translational Research Section.
“Our approach will help scale up manufacturing and will speed delivery of tissues to the clinic,” added Bharti, who led the research along with Carl Simon Jr. and Peter Bajcsy of NIST.
Cells of the RPE nourish the light-sensing photoreceptors in the eye and are among the first to die from geographic atrophy, commonly known as “dry” AMD. Photoreceptors die without the RPE, resulting in vision loss and blindness.
Bharti’s team is working on a technique for making RPE replacement patches from AMD patients’ own cells. Patient blood cells are coaxed in the lab to become induced pluripotent stem cells (iPSCs), which can become any type of cell in the body. The iPSCs are then seeded onto a biodegradable scaffold where they are induced to differentiate into mature RPE. The scaffold-RPE “patch” is implanted in the back of the eye, behind the retina, to rescue photoreceptors and preserve vision.
The patch worked in an animal model, and a clinical trial is planned.
The researchers’ AI-based validation method used deep neural networks, an AI technique that performs mathematical computations aimed at detecting patterns in unlabeled and unstructured data. The algorithm operated on images of the RPE obtained using quantitative bright-field absorbance microscopy. The networks were trained to identify visual indications of RPE maturation that correlated with positive RPE function.
Those single-cell visual characteristics were then fed into traditional machine-learning algorithms, which in turn helped the computers learn to detect discrete cell features crucial to the prediction of RPE tissue function.
The method was validated using stem cell–derived RPE from a healthy donor. Its effectiveness was then tested by comparing iPSC-RPE derived from healthy donors with iPSC-RPE from donors with oculocutaneous albinism disorder and with clinical-grade stem cell–derived RPE from donors with AMD.
In particular, the AI-based image-analysis method accurately detected known markers of RPE maturity and function: transepithelial resistance, a measure of the junctions between neighboring RPE; and secretion of endothelial growth factors. The method also can match a particular iPSC-RPE tissue sample to other samples from the same donor, which helps confirm the identity of tissues during clinical-grade manufacturing.
“Multiple AI methods and advanced hardware allowed us to analyze terabytes and terabytes of imaging data for each individual patient and do it more accurately and much faster than in the past,” Bajcsy said.
“This work demonstrates how a garden-variety microscope, if used carefully, can make a precise, reproducible measurement of tissue quality,” Simon said.
The work was supported by the NEI Intramural Research Program and the Common Fund Therapeutics Challenge Award. The flow-cytometry core, led by the National Heart, Lung and Blood Institute, also contributed to the research. (NIH authors: N.A. Hotalin, Q. Wan, R. Sharma, A. George, and K. Bharti, J Clin Invest, 2019; DOI:10.1172/JCI131187)
This page was last updated on Tuesday, March 29, 2022
