Research Briefs
NIDDK: FIRST PHOTOACTIVE DRUG TO FIGHT PARKINSON DISEASE
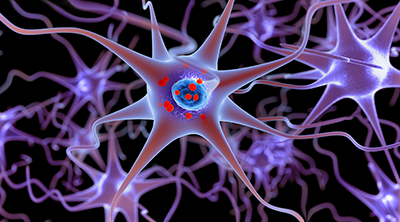
CREDIT: LARS NEUMANN, THINKSTOCK.COM
An NIDDK researcher was part of an international team that designed the first potentially therapeutic photoactive drug—MRS7145—to fight Parkinson disease. Shown: A 3-D illustration showing neurons containing Lewy bodies (small red spheres), which are deposits of alpha-synuclein proteins accumulated in the brain cells.
An NIDDK researcher was part of an international team that designed the first potentially therapeutic photoactive drug—MRS7145—to fight Parkinson disease (PD), a neurodegenerative disorder caused by a loss of dopamine, which in turn causes a gradual loss of motor control. Although there is no cure, there are drugs (such as levodopa) and treatments (such as deep-brain stimulation) to treat symptoms, which include tremors, stiffness, slowed body movements, and poor balance. But the action of conventional drugs can be limited because they can’t precisely target the areas of the brain involved in PD, their effects diminish over time and the dosage may have to be increased, and newer drugs have unwanted side effects such as uncontrolled body movements. But scientists have found that they can use certain wavelengths of light to control the precision of light-sensitive drugs and thereby avoid adverse effects. The researchers in the current study developed MRS7145, a photo-sensitive adenosine receptor (A2AR) antagonist. A2A receptors play a role in controlling movement. The MRS7145 is activated by violet light in a wavelength that is non-toxic to neurons. In this study, which involved rodent models of PD, the researchers implanted optical fibers in the striatum (area of the brain that controls motor activity), then used a remote-control device to trigger the release of light. The light activated the MRS7145, which blocked the A2A receptors, allowing dopamine to be released and reducing the PD symptoms. Although this drug has only been tested in laboratory animals so far, the researchers hope their work will lead to new therapeutic solutions for treating PD. (NIDDK researcher: K. Jacobson, J Control Release, 283:135-142, 2018, DOI: 10.1016/j.jconrel.2018.05.033)
NICHD: POSSIBLE LINK BETWEEN GESTATIONAL DIABETES AND EARLY-STAGE KIDNEY DAMAGE
Researchers may have uncovered a potential link between women who have had gestational diabetes and early stage kidney damage. According to the study led by NICHD investigators and other institutions, women who had gestational diabetes were more likely to have a high glomerular filtration rate (GFR), which is an estimate of how much blood passes through the glomeruli (the tiny filters within the kidneys that extract the waste) per minute. Researchers believe that a very high GFR rate can be a sign of early kidney damage. The work was conducted as part of the NICHD-funded Diabetes and Women’s Health Study. Researchers collected and analyzed blood and urine samples from Danish women who had pregnancies from 1996 through 2002. The data included results from tests for diabetes and kidney functioning an average of 13 years later. The group included 601 women who had gestational diabetes and 613 who did not. Results showed that women who had gestational diabetes and later developed diabetes were approximately nine times more likely to have an elevated GFR later in life compared to women who did not have gestational diabetes. In addition, women who had gestational diabetes and later-life diabetes were also more likely to have elevated urinary albumin to creatine ratio (UACR), an indicator of kidney disease. Although the study could not prove that gestational diabetes is a direct cause of kidney damage, more research needs to be conducted in order to confirm these findings. The authors noted, however, that women who have had gestational diabetes may benefit from periodic checkups to monitor for early-stage renal damage so clinicians can initiate treatment to prevent or delay further disease progression. (NIH Authors: S. Rawal, S.N. Hinkle, J. Wu, E. Yeung, J.L. Millis and C. Zhang, Diabetes Care 41:1378-1384; DOI 10.2337/dc17-2629)
NCI: ENHANCING TUMOR RESPONSE TO RADIOTHERAPY
Radiotherapy plays a significant role in the management of non–small cell lung cancer (NSCLC), but recurrence after treatment remains problematic. In a recent study, investigators at NCI found a novel combination-therapy approach to treat NSCLC. The researchers used a CDK4/6 inhibitor, abemaciclib, and combined it with ionizing radiation (IR). CDK4/6 inhibitors are a class of drugs designed to interrupt the growth of cancer cells. Abemaciclib, a drug used to treat advanced metastatic breast cancer, was combined with IR to enhance radiosensitivity of NSCLC cells and tumor xenografts in mice. The drug inhibited phosphorylation of the retinoblastoma (RB) protein and effectively blocked cell proliferation in cells as well as in mouse tumors. This combination therapy resulted in significant inhibition of IR-induced DNA-damage repair. Several NSCLC cell lines with varied genetic backgrounds were tested for the effect of this novel drug and IR combination. The study identified enhanced radiation-induced cell killing with this combination to be more effective in cells with functional RB protein. Interestingly, in this study the researchers found an unexpected role of abemaciclib in inhibiting radiation-induced vasculogenesis (the process of blood vessel formation). Tumor vasculogenesis is a backup pathway to restore tumor vasculature that may contribute to recurrence of a tumor after radiotherapy. The study found that abemaciclib, when administered during and after IR treatment, inhibited vasculogenesis thus delaying tumor regrowth. Abemaciclib unexpectedly inhibited vasculogenesis by reducing the activation of tumor hypoxia inducible factor-1 and the recruiting of bone-marrow-derived immune cells to the tumor. The findings suggest that abemaciclib in combination with IR could provide a novel biomarker-driven combination therapeutic strategy for patients with NSCLC. (NCI authors: S. Naz, A.L. Sowers, R. Choudhuri, M.F. Wissler, J. Gamson, A. Mathias, J.A. Cook, J.B. Mitchell, Clin Cancer Res, DOI: 10.1158/1078-0432.CCR-17-3575; 2018)
NIEHS, NCI: OBESITY ASSOCIATED WITH LOWER BREAST CANCER RISK IN YOUNG WOMEN

Young women with high body fat have a decreased chance of developing breast cancer before menopause, according to NIH scientists and an international team of researchers in the Premenopausal Breast Cancer Collaborative Group. The international team pooled data from 19 different sites comprising more than 750,000 women (ranging in age from 18 to 54) from around the world. The researchers evaluated questionnaires, filled out by study volunteers, that included height, weight, and other health-related factors. It was determined that the relative risk of premenopausal breast cancer was reduced 12 to 23 percent for each five-unit increase in body mass index (BMI). The strongest effect was seen in relation to BMI at ages 18-24, with very obese women in this age group being 4.2 times less likely to develop premenopausal breast cancer compared to women with low BMI at the same age. The NIH researchers are unsure why young, premenopausal women with a high BMI appear to be protected against breast cancer and caution that young women should not intentionally gain weight to lower breast-cancer risk as there are so many health risks associated with being overweight or obese. Understanding the biological mechanisms underlying the association of BMI and the risk of breast cancer could have important implications for breast-cancer prevention. (NIH authors: D.P. Sandler, C.M. Kitahara, M.S. Linet, K.M. O’Brien, JAMA Oncol; DOI: 10.1001/jamaoncol.2018.1771)
NCATS, NCI: POTENTIAL NEW APPROACH TO STOP CANCER METASTASIS
NIH researchers and collaborators at Northwestern University (Evanston, Illinois) have identified a compound that blocks the spread of pancreatic and other cancers in various animal models. The team identified a compound, which they named metarrestin, that stopped tumor metastasis in multiple animal models. Mice treated with metarrestin also had fewer tumors and lived longer than mice that did not receive treatment. Metarrestin breaks down an incompletely understood component of cancer cells called the perinucleolar compartment (PNC). PNCs are found only in cancer cells, and in a higher number of cells in advanced cancer when it has spread to other sites in the body. The Northwestern scientists had shown earlier that the more cancer cells with PNC in a tumor, the more likely it would spread. Those findings suggested that reducing PNCs might translate to less cancer progression and better outcomes in patients. To test these ideas, the Northwestern scientists relied on NCATS’s expertise in screening, chemistry, and compound development and testing to evaluate more than 140,000 compounds for their potential effectiveness in eliminating PNCs in cells in advanced cancer. The investigators identified one compound, metarrestin, that could effectively break down PNCs in advanced prostate cancer cells. The NCI researchers evaluated the effects—including toxicity—of metarrestin in pancreatic cancer mouse models. The scientists found that the compound prevented the further spread of pancreatic cancer by disrupting the protein-making machinery of cancer cells, and that mice treated with metarrestin lived longer than mice without treatment. The NCI and NCATS scientists are working together to collect the preclinical data on metarrestin needed to further its development as a candidate drug. The scientists plan to file an Investigational New Drug (IND) application in the fall with the FDA. FDA IND approval is necessary before a candidate drug can be tested in patients. (NIH authors: Udo Rudloff (NCI), Juan Jose Marugan (NCATS), and 15 others, Sci Trans Med 10:eaap8307, 2018; DOI: 10.1126/scitranslmed.aap8307)
NCCIH, NIBIB: OPIOID THERAPY MAY NOT ALWAYS WORK WELL FOR CHRONIC PAIN
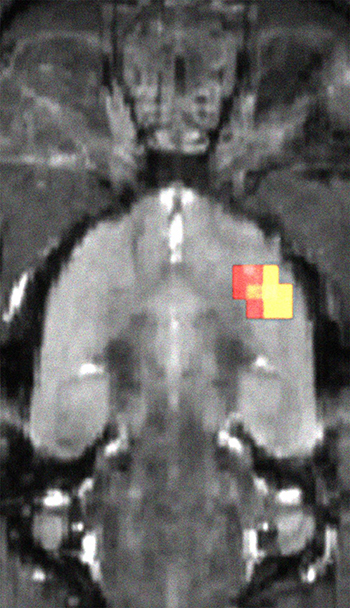
CREDIT: SCOTT J. THOMPSON, NIAID
Brain regions showing less opioid receptor availability.
Researchers have shown that pain-induced changes in the rat brain’s opioid receptor system may explain the limited effectiveness of opioid therapy in chronic pain and may play a role in the depression that often accompanies it. These findings clearly show the impact of chronic pain on the brain and its relation to depression. In the study, cross-sectional positron emission tomography (PET) imaging was performed on the brains of 17 rats that had undergone surgery to produce a nerve injury that causes chronic pain and on 17 rats that had undergone sham surgery. Three months later, the availability of opioid receptors had decreased in multiple regions of the brain in the nerve-injured rats, but no changes had occurred in the sham-surgery rats. Other tests performed on the rats provided insight into the relationship between chronic pain and depression. Normal, healthy rats will drink water with sucrose (sugar) in it rather than plain water if given a choice. But animals with a decreased ability to experience pleasure, a recognized symptom of depression known as anhedonia, may not. In this study, the rats with chronic pain showed a decreased preference for sucrose water over plain water after injury; furthermore, animals in this group also displayed a positive correlation between opioid receptor availability and the preference for sucrose water. Although this study suggests that neuropathic pain-evoked changes in cerebral opioid receptor expression may impact depression, human studies are needed to confirm these findings. (NIH authors: S.J. Thompson, M.H. Pitcher, F. Tarum, G. Niu, X. Chen, D.O. Kiesewetter, M.C. Bushnell, Pain DOI: 10.1097/j.pain.0000000000001282)
NIAID: BACTERIA THERAPY FOR ECZEMA SHOWS PROMISE
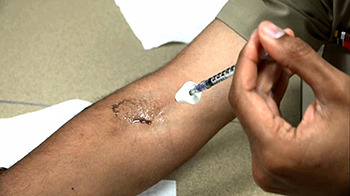
CREDIT: NIAID
A scientist demonstrates application of the experimental therapy to the inner elbow. For demonstration purposes, the bacteria solution has been replaced with purple dye.
Topical treatment with live Roseomonas mucosa—a bacterium naturally present on the skin—was safe for adults and children with atopic dermatitis (eczema) and was associated with reduced disease severity, according to initial findings from an ongoing early-phase clinical trial at NIAID. Preclinical work in a mouse model of atopic dermatitis had suggested that R. mucosa strains collected from healthy skin can relieve disease symptoms. The new findings support further evaluation of this potential new therapy.
The cause of atopic dermatitis is unknown, but studies suggest that the skin microbiome plays a key role. For years, scientists have known that people with atopic dermatitis tend to have large populations of Staphylococcus aureus bacteria on their skin. These bacteria can cause skin infections and trigger immune responses that increase inflammation and worsen symptoms.
Recent work by NIAID researchers using mouse and cell culture models of atopic dermatitis revealed that treatment with isolates of R. mucosa collected from the skin of healthy people improved disease outcomes in the models. In contrast, R. mucosa isolates from people with atopic dermatitis either had no impact or worsened outcomes in the models.
Based on these preclinical findings, NIAID investigators designed an early-stage clinical trial to test the safety and potential benefit of a treatment containing live R. mucosa in people with atopic dermatitis. Final results from the ongoing study at NIH will provide the foundation for larger trials to evaluate the efficacy of this novel investigational therapy, as well as to better understand the role of R. mucosa in atopic dermatitis. NIH has exclusively licensed the technology to Forte Biosciences to advance this potential new therapy through further clinical development. (NIH authors: I.A. Myles, N.J. Earland, E.D. Anderson, I.N. Moore, M.D. Kieh, K.W. Williams, A. Saleem, N.M. Fontecilla, P.A. Welch, D.A. Darnell, L.A. Barnhart, A.A. Sun, G. Uzel,and S.K. Datta, JCI Insight 3:e120608, 2018)
NIMH: BIGGER HUMAN BRAIN PRIORITIZES THINKING HUB – AT A COST
Some human brains are nearly twice the size of others–but how might that matter? NIMH researchers and their NIH grant-funded colleagues have discovered that these differences in size are related to the brain’s shape and the way it is organized. The bigger the brain, the more its additional area is accounted for by growth in thinking areas of the cortex—at the expense of relatively slower growth in lower order emotional, sensory, and motor areas. The study incorporated brain scan data from more than 3,000 people. To pinpoint how the human brain’s organization varies in relation to how big it is, the researchers analyzed magnetic resonance imaging brain scans of youth from the Philadelphia Neurodevelopmental Cohort, an NIMH IRP sample, and the Human Connectome Project.
Since people with certain mental disorders show alterations in brain size related to genetic influences, the new cortex maps may improve an understanding of altered brain organization in disorders. The higher expanding regions are also implicated across diverse neurodevelopmental disorders, so the new insights may hold clues to understanding how genetic and environmental changes can impact higher mental functions. (NIH authors: P.K. Reardon, J. Seidlitz, S. Liu, L.S. Clasen, J.D. Blumenthal, F.M. Lalonde, A. Raznahan, Science eaar2578, DOI: 10.1126/science.aar2578)
NIAID: MICROGLIA ARE KEY DEFENDERS AGAINST PRION DISEASES
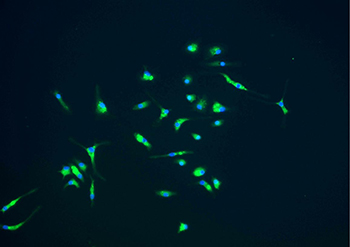
CREDIT: NIAID
Microglia, shown in green, are part of the immune response that protect the brain. They could play a role in slowing the progress of prion and other neurodegenerative diseases.
NIAID scientists have used an experimental drug, PLX5622, to test the role of microglia against scrapie, a prion disease of sheep. PLX5622 rapidly kills most of the microglia in the brain. When researchers gave the drug to mice infected with scrapie, microglia were eliminated and the mice died one month faster than did untreated mice. The results suggest that microglia can defend against a prion infection and thus slow the course of disease. The scientists hypothesize that microglia trap and destroy the aggregated prion proteins that cause brain damage; are now studying how microglia may be able to destroy prions in the brain; and note that microglia could have a similar beneficial effect on other neurodegenerative diseases associated with protein aggregation, such as Alzheimer disease and Parkinson disease. (NIH authors: J.A. Carroll, B. Race, K. Williams, J. Striebel, and B. Chesebro, J Virol DOI:10.1128/JVI.00549-18)
This page was last updated on Thursday, April 7, 2022
