The SIG Beat: Periodontitis
NEWS FROM AND ABOUT THE SCIENTIFIC INTERESTS GROUPS
Periodontitis: A Microbial-Driven Inflammatory Disease
A mouth microbiome that’s out of whack can lead to serious health problems such as the chronic inflammatory disease periodontitis. Triggered by bacterial biofilms, periodontal disease causes inflammation that damages gum tissue and can destroy the bone that supports the teeth. According to the CDC, over 47 percent of American adults over 30 have mild, moderate, or severe forms of the disease. If left untreated, periodontitis can lead to tooth loss and may contribute to other inflammatory diseases such as diabetes and heart disease. More research is needed, however, to clarify the relationship between gum disease and health problems beyond the mouth.
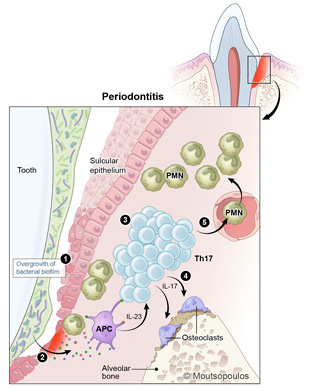
CREDIT: ALAN HOOFRING, NIH MEDICAL ARTS
In periodontitis, an imbalanced microbiome triggers an exaggerated IL-17 immune response that leads to the activation of osteoclasts (cells that degrade bone) and bone loss. The overgrowth of bacterial biofilm stimulates antigen-presenting cells (APC) to upregulate the cytokine IL-23, which stimulates the development of T-helper (Th17) cells. Th17 cells produce the cytokine IL-17 and activate osteoclasts to resorb bone. IL-17 will also stimulate the recruitment of polymorphonuclear neutrophils (PMN) to the area, which will further amplify immunopathology in the lesion of periodontitis.
National Institute of Dental and Craniofacial Research Clinical Investigator Niki Moutsopoulos is conducting research on periodontitis in order to understand its mechanisms and explore possible therapies. She described her work at a seminar hosted by the Inflammatory Disease Scientific Interest Group (SIG) on January 19, 2016.
To determine how immune defects may be associated with susceptibility to periodontal disease, Moutsopoulos recruited healthy volunteers with and without periodontitis and people with the monogenic immune defect leukocyte-adhesion deficiency (LAD-I). Her research team did clinical phenotyping, microbiome characterization, molecular profiling, and in vitro assays with human cells. She used periodontal probes to measure the loss of tooth-supporting structures and found that people with LAD-I had both increased bone loss and increased bacterial biomass on the surfaces of their teeth. Furthermore, the expression of the protein integrin beta-2 (CD18) was inversely correlated with periodontitis severity, suggesting a link between defective neutrophil migration and the severity of LAD-I periodontitis.

Niki Moutsopoulos is conducting research on periodontitis in order to understand its mechanisms and explore possible therapies.
The team also compared cytokine and chemokine gene expression in LAD-I patients to that of healthy people who had severe to mild periodontitis. Interestingly, the cytokines interleukin-23 (IL-23) and IL-17 were induced in LAD-I periodontitis. Another finding was that the LAD-I microbiome stimulated an IL-17 immune response. It was also determined that abnormal neutrophil recruitment caused the upregulated IL-17 inflammatory response in the periodontal tissue and bone loss associated with bacterial overload. Finally, preliminary work using the drug ustekinumab (Stelara) to inhibit IL-23 is showing promise in patients.
Inflammation is a complicated biological response to injury or harmful stimuli such as invading pathogens. Inflammatory responses can be protective when they are working properly, but their dysregulation or persistence can be destructive.
The Inflammatory Disease SIG aims to bring together scientists to encourage discussions and NIH-wide collaborations that could potentially develop new treatments for inflammatory diseases. The SIG will host bimonthly seminars and symposiums that focus on the basic and translational characteristics of inflammation. To join the LISTSERV (INFLAM-DIS-L), visit https://list.nih.gov/cgi-bin/wa.exe?SUBED1=INFLAM-DIS-L&A=1 or contact Thomas A. Wynn at twynn@niaid.nih.gov.
This page was last updated on Thursday, April 14, 2022
