Research Briefs
NIDDK, NIAID, CC, NICHD: INCREASED CORONARY-VESSEL WALL THICKNESS IN HIV-INFECTED YOUNG ADULTS
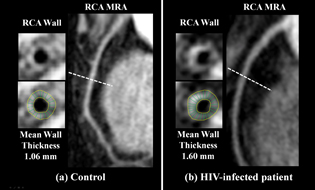
KHALED ABD-ELMONIEM, COLLEEN HADIGAN, AHMED M. GHARIB, NIDDK
Time-resolved acquisition of phase-sensitive dual-inversion recovery (TRAPD) vessel-wall magnetic resonance imaging of the proximal right coronary artery (RCA) in a control subject (a) and a person infected with HIV (b). Shown on the left side of each panel are the TRAPD RCA wall image (top) and the calculation of the mean wall thickness (bottom). Slices were acquired in the proximal segment of the RCA as shown on the corresponding coronary magnetic resonance angiography (MRA) image on the right side of each panel (dotted lines).
Individuals with long-term human immunodeficiency virus (HIV) infection are at risk for premature vasculopathy and cardiovascular disease (CVD), according to an NIH intramural study: a prospective cross-sectional study of 35 young adults who acquired HIV in early life and 11 healthy control subjects, free of CVD. The researchers used time-resolved acquisition of phase-sensitive dual-inversion recovery (TRAPD) coronary vessel-wall magnetic resonance imaging to measure proximal right coronary artery (RCA) wall thickness; they used multidetector computed-tomography angiography to measure coronary plaque and epicardial fat. They found that the RCA vessel-wall thickness was significantly higher in HIV-infected patients than in control subjects. However, the amount of plaque was not sufficient to cause luminal narrowing; plaque was not related to RCA wall thickness; and the epicardial fat did not differ between groups. Increased duration of antiretroviral therapy, hyperlipidemia, and smoking contributed to the thickening of the coronary wall, independent of atherosclerotic plaque. These modifiable risk factors appear to influence early atherogenesis as measured by coronary wall thickness and may be important targets for CVD risk reduction. (NIH authors: K.Z. Abd-Elmoniem, A.B. Unsal, S. Eshera, J.R. Matta, N. Muldoon, D. McAreavey, J.B. Purdy, R. Hazra, C. Hadigan, and A.M. Gharib, Clin Infect Dis 15:1779–1786, 2014)
NICHD: SOME SUNSCREEN CHEMICALS MAY REDUCE FERTILITY IN MEN
Some sunscreen chemicals used to protect against ultraviolet (UV) rays may impair men’s ability to father children, according to a study by NICHD and the New York State Department of Health’s Wadsworth Center. Benzophenone (BP)-type UV filters comprise a class of about 29 chemicals commonly used in sunscreens and other personal-care products to protect skin and hair from sun damage. Some of these chemicals, when absorbed by the skin, can interfere with the body’s endocrine system. In a study with 501 couples who were part of the Longitudinal Investigation of Fertility and the Environment Study, researchers found that men with high exposure to UV filters 2,2',4,4'-tetrahydroxybenzophenone (BP-2) or 4-hydroxybenzophenone (4OH-BP) had a 30 percent reduction in fecundity, the biological ability to reproduce. Lower fecundity may result in a longer time to pregnancy. The researchers caution that the results are preliminary and that additional studies are needed to confirm their findings. (NICHD authors: G.M. Buck Louis, K. Kannan, K.J. Sapra, J. Maisog, R. Sundaram; Am J Epidemiol 180:1168–1175, 2014)
NICHD: CHRONIC HIGH BLOOD GLUCOSE MAY BE DETRIMENTAL TO THE DEVELOPING BRAIN OF YOUNG CHILDREN
Young children who have long-term high blood glucose concentrations are more likely to have slower brain growth, according to NICHD and other researchers. Although there were no significant cognitive differences between the healthy children and those with type 1 diabetes, the researchers believe a continuing study with the same groups of children may show changes in that respect as well. The findings could lead to a major shift in the way children with type 1 diabetes are treated. The study tracked 144 children ages 4 to 9 who had been living with type 1 diabetes for an average of two and one-half years. Researchers used magnetic resonance imaging scans to examine brain structure and function in these children compared with 72 healthy children, including siblings. The children were well matched for age, body mass index, and socioeconomic status. Researchers found significantly slower brain growth overall as well as in specific brain regions in children with diabetes. Traditionally, pediatricians have allowed young children with type 1 diabetes to maintain above-normal blood glucose concentrations. The theory was that it was safer to run high than low, because consistently low blood glucose concentrations could raise the risk of a child having a seizure. This study shows that chronic high blood glucose may slow growth in the brain’s gray matter, which affects cells and signals, as well as in the brain’s white matter, which affects the brain’s wiring. In addition to high blood glucose concentrations, researchers found that wide swings in blood glucose, both spiking too high and dropping too low. These sharp swings raised concerns about the effect on brain function. (NICHD author: K. Winer; Diabetes DOI:10.2337/db14-1445)
NIAID: NIAID-GSK EXPERIMENTAL EBOLA VACCINE APPEARS SAFE, STIMULATES IMMUNE RESPONSE
An experimental vaccine to prevent Ebola virus disease was well-tolerated and produced immune system responses in all 20 healthy adults who received it in a phase 1 clinical trial conducted by NIH researchers. The candidate vaccine, which was co-developed by NIAID’s Vaccine Research Center (VRC) and GlaxoSmithKline (GSK), was tested at the Clinical Center. The investigators also analyzed the research participants’ blood to learn whether the vaccine prompted the production of T cells. A recent study, published in Nature Medicine, by VRC scientist Nancy J. Sullivan and colleagues, showed that nonhuman primates inoculated with the candidate NIAID-GSK vaccine developed both antibody and T-cell responses, and that these responses were sufficient to protect vaccinated animals from disease when they were later exposed to high amounts of Ebola virus. In the clinical trial, the experimental NIAID-GSK vaccine induced a T-cell response in many of the volunteers, including production of CD8 T cells, which may be an important part of immune protection against Ebola viruses. Four weeks after vaccination, CD8 T cells were detected in two volunteers who had received the lower-dose vaccine and in seven of those who had received the higher dose. There were no serious adverse effects observed in any of the volunteers, although two people who received the higher-dose vaccine did develop a brief fever within a day of vaccination. Additional details about this trial, VRC 207, are available at http://www.clinicaltrials.gov using the identifier NCT02231866. For more information about early-stage Ebola vaccine clinical trials, see http://www.niaid.nih.gov/news/QA/Pages/EbolaVaxResultsQA.aspx. (Clinical trial: VRC/NIAID authors: J. E. Ledgerwood et al., N Engl J Med DOI:10.1056/NEJMoa1410863; Sullivan study: VRC authors: D.A. Stanley, N.J. Sullivan, et al., Nat Med 20:1126–1129, 2014)
NIDA: NIDA RESEARCHERS CONFIRM IMPORTANT BRAIN-REWARD PATHWAY
A NIDA study showed that stimulation of glutamate neurons in a specific brain region (the dorsal raphe nucleus) leads to activation of dopamine-containing neurons in the dopamine reward system. Drug-abuse researchers have long been interested in the dorsal raphe nucleus because nerve cells in this area connect to part of the dopamine reward system. Many of the pathways are rich in serotonin, a neurotransmitter linked to mood regulation. Even though electrical stimulation of the dorsal raphe nucleus promotes reward-related behaviors, drugs that increase serotonin have low abuse potential. This finding has led researchers to theorize that another neurotransmitter may be responsible for the role that the dorsal raphe nucleus plays in reward. In these rodent models, researchers used special tracers and labeling compounds to confirm that this circuit begins with glutamate cells in the dorsal raphe nucleus that connect to dopamine cells in the ventral tegmental area, signaling in turn travel the nucleus accumbens, a brain structure linked to motivation, pleasure, and reward. After verifying the pathway, investigators used optogenetic techniques and chemical blockers to confirm that glutamate, not serotonin, is responsible for activating this reward circuitry. The discovery of this pathway opens new avenues for examining its participation in a variety of disorders related to motivation. (NIDA authors: A. Buendia, A.F. Hoffman, C.R. Lupica, and M. Morales, Nat Commun DOI:10.1038/ncomms6390)
NIEHS: NIH SCIENTISTS DETERMINE HOW ENVIRONMENT CONTRIBUTES TO SEVERAL HUMAN DISEASES
Using a new imaging technique, NIEHS researchers have found that the biochemical machinery that synthesizes DNA can insert into the DNA strand molecules that are damaged as a result of environmental exposures. These damaged molecules trigger cell death, which can produce some human diseases. The work provides a possible explanation for how one type of DNA damage may lead to cancer, diabetes, hypertension, cardiovascular and lung disease, and Alzheimer disease. The researchers used time-lapse crystallography to determine that DNA polymerase, the enzyme responsible for assembling the normal nucleotides of DNA, also incorporates damaged nucleotides into the DNA strand. Time-lapse crystallography is a technique that takes snapshots of biochemical reactions occurring in cells. The damage to the nucleotides is caused by oxidative stress, or the generation of free oxygen molecules, in response to environmental factors, such as ultraviolet exposure, dietary factors, and chemical compounds found in paints, plastics, and other consumer products. Scientists suspected that the DNA polymerase was inserting nucleotides that were damaged by carrying an additional oxygen atom. When one of these oxidized nucleotides is placed into the DNA strand, it can’t pair with the opposing nucleotide as usual, which leaves a gap in the DNA. Until this study, no one had actually seen how the polymerase did it or understood the downstream implications. The scientists stressed that the quantities of oxidized nucleotides in the nucleotide pool are usually under tight control, so if they accumulate and start to outnumber undamaged nucleotides, the DNA polymerase adds more of them to the strand. Molecules that inhibit oxidation, known as antioxidants, reduce the number of oxidized nucleotides and may help prevent some diseases. (NIEHS authors: B.D. Freudenthal, W.A. Beard, L. Perera, D.D. Shock, and S.H. Wilson, Nature DOI:10.1038/nature13886)
This page was last updated on Tuesday, April 26, 2022
