NCI Campus in Frederick
What’s Happening in NCI’s Storied Labs to the North?
NIH Catalyst readers might be aware that the National Cancer Institute (NCI) has research labs in Frederick, Md. But how many know that—or why, for that matter—those labs are inside the gates of an army base? Or that what used to be called simply NCI-Frederick is now a composite of a recently designated national lab, several NCI Center for Cancer Research (CCR) intramural labs, and an administrative entity called the NCI Campus at Frederick?

This article highlights changes that have taken place at Frederick and offers a peek into the important intramural research and other collaborations now conducted in the NCI and contractor labs there.
The NCI has a 40-year relationship with the city of Frederick, Md., a Civil War–era mining and farming hub still surrounded by ample farmland some 35 miles north of the Bethesda campus NCI labs. In 1971, President Richard Nixon requested that approximately 70 acres and 60 buildings belonging to the U.S. Army at Fort Detrick be “converted into a leading center for cancer research” to be led by the NCI in partnership with the private sector.
The transfer met two of Nixon’s goals: the termination of research on offensive biological weapons, which took place at Fort Detrick, and the campaign to eradicate cancer. Nixon wrote in a presidential statement in October 1971 that “some of the Nation’s most sophisticated scientific facilities” were at Fort Detrick and that they could be “converted so effectively and so inexpensively to an intensive program of cancer research.”
The U.S. Department of Health and Human Services (HHS) quickly established the National Cancer Research and Development Center with only a few dozen employees. By 1973 the burgeoning facility became the Frederick Cancer Research and Development Center. In 1975, the facility was designated as a Federally Funded Research and Development Center (FFRDC)—a government-owned, contractor-operated enterprise—analogous to the famed Los Alamos or Lawrence Livermore national labs.
The FNL has had several names through the years, such as Frederick Cancer Research Center dating to 1981, still seen in some signs around the campus, and the ever-colloquial NCI-Frederick.
In March 2012, the FFRDC portion of NCI-Frederick was designated the Frederick National Laboratory for Cancer Research (FNLCR). Of the nation’s 39 national labs, FNLCR is the only one owned by HHS and devoted exclusively to biomedical research and development. FNLCR’s largest contractor is SAIC-Frederick (Science Applications International Corporation), which won its first contract from NCI in 1995.
The NCI organization at Frederick may seem a bit complicated to understand. Think of it as a sort of island campus within Fort Detrick and around the Frederick area. On that island are labs and offices belonging to the FNLCR (owned by NCI and operated by SAIC); the NCI Campus at Frederick administrative offices; and about 10 out of 50 of NCI’s Center for Cancer Research (CCR) labs. The other CCR labs are located at the NIH Bethesda campus. The FNLCR also includes facilities throughout the region, such as the new Advanced Technology Research Facility (ATRF) in Frederick. For an organizational diagram, see http://ncifrederick.cancer.gov/About/AtAGlance.aspx.
The FNLCR has “a very broad mission, which is valuable to many people across the nation,” said Craig Reynolds, the director of the NCI Office of Scientific Operations in Frederick. Its activities and resources are not only available for NCI and other NIH intramural researchers but also “for academia, industry, and other government agencies in the country and around the world.” That mission includes biomedical research on cancer as well as on AIDS and other infectious diseases.
And so, with that background, this article highlights some of the scientific programs and biomedical findings assciated with NCI on the Frederick campus.
FNLCR Programs
FNLCR researchers work in concert with government colleagues and external collaborators to advance research in cancer and AIDS, identify unmet needs, develop novel experimental approaches, and implement new technologies.
Biopharmaceutical Development Program
The Biopharmaceutical Development Program (BDP), established in 1993, is funded by NCI’s Division of Cancer Treatment and Diagnosis (DCTD) and supports the development of biopharmaceuticals, including monoclonal antibodies, recombinant proteins, therapeutic peptides and DNA vaccines, virus therapeutics and vaccines, gene-therapy products, and other biological agents. The program aligns closely with DCTD’s mission to support the translation of promising research into improved diagnostic and therapeutic treatments for cancer patients. One of BDP’s early projects in the 1990s was producing monoclonal antibodies—when no pharmaceutical company would—to treat children with neuroblastoma, a common childhood cancer that grows in parts of the nervous system. BDP’s resources are available to intramural and extramural NIH investigators, government agencies, and independent parties. For more information visit http://ncifrederick.cancer.gov/Programs/Science/BDP.
Center for Advanced Preclinical Research
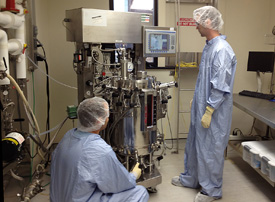
VINAY VYAS
In the BDP lab: From left, Darren Benedick and Mark Slatcoff set up the 80-L bioreactor for production of recombinant human interleukin-15.
NCI’s Center for Advanced Preclinical Research (CAPR), which is funded by NCI-CCR and operated by the FNLCR, is developing a comprehensive preclinical trial framework for evaluating the anti-tumor efficacy and selectivity, biodistribution, and metabolism of early-stage candidate drugs using genetically engineered mouse models. CAPR serves as a national resource for the comprehensive preclinical testing of early-stage candidate drugs. Candidate compounds are assessed for anti-tumor efficacy and selectivity in genetically engineered animal models. For more information, visit http://atp.ncifcrf.gov/atpi/ppt/capr. Today, intramural researchers in NCI-CCR’s HIV Drug Resistance Program Retroviral Replication Laboratory are continuing to obtain a better understanding of important events in the life cycle of human retroviruses, especially HIV.
HIV and AIDS
In the 1980s, the human immunodeficiency virus (HIV) was identified as the cause of AIDS. But for pharmaceutical companies to develop a diagnostic test for HIV, large amounts of the virus needed to be grown quickly. Luckily, NCI’s Frederick laboratories already had a facility for producing retroviruses that cause cancer and it refocused its efforts to produce large amounts of HIV instead. NCI partnered with five private companies and provided them HIV viruses, and within a year the effort produced an FDA-approved blood test to detect HIV. The use of diagnostic tests to detect and discard blood from infected individuals resulted in the rapid decrease of blood transfusion–associated infections in the United States.
Today, researchers in FNLCR’s AIDS and Cancer Virus Program along with intramural researchers in CCR’s HIV Drug Resistance Program Retroviral Replication Laboratory are continuing to obtain a better understanding of important events in the life cycle of human retroviruses, especially HIV.
FNLCR’s New Building

Staff photo, Scientific Publications, Graphics & Media, SAIC-Frederick, Inc.
FNLCR opened a new building—the Advanced Technology Research Facility (ATRF)—in 2012 that consolidates labs and operations that had been scattered among more than 30 buildings at NCI’s Frederick campus.
In 2012, FNLCR opened a new building—the Advanced Technology Research Facility (ATRF)—that consolidates labs and operations that had been scattered among more than 30 buildings at NCI’s Frederick campus. The ATRF houses laboratories of FNLCR’s Advanced Technology Program and the Biopharmaceutical Development Program and also dedicates space to accommodate co-location partnerships. It is available for cooperative research and development projects with academia, government, and life-sciences industry partners to accelerate cancer and AIDS translational research. For more information, visit http://frederick.cancer.gov/Partnerships/Atrf.aspx. For more information about the Frederick National Lab, go to http://frederick.cancer.gov.
Intramural Research in Frederick
NCI’s Center for Cancer Research (CCR)—part of NCI’s intramural research program—maintains a research program on the Frederick Campus. Out of CCR’s 50-some branches, programs, and labs, more than 10 are in Frederick, accounting for 30 percent of CCR’s principal investigators. For a complete list and description of the CCR labs that are located in Frederick, go to http://frederick.cancer.gov/Science/NciAtFNLCR.aspx. Following is a sampling.
Mouse Cancer Genetics Program
The CCR Mouse Cancer Genetics program, established in 1999, uses mouse molecular genetics to understand the fundamental processes underlying mammalian development and diseases. Major areas of focus include developmental mechanisms and pathways usurped in cancer, stem cell and cancer-propagating cell function, genome integrity control, tumor suppressor function, neurotrophin-activated signaling pathways and angiogenesis.
Molecular Targets Laboratory
Recent advances and insights into the molecular pathogenesis of cancer provide unprecedented opportunities for discovery and development of novel, molecularly targeted diagnostic, therapeutic and preventative strategies and agents. The CCR’s Molecular Targets Laboratory (MTL) facilitates the discovery of compounds—synthetic as well as natural products—that may serve as bioprobes for functional genomics, proteomics and molecular target validation research, as well as candidates for drug development.

Some 200,000 extracts from terrestrial and marine species around the globe, such as the sponge Phallusia julinea, have been collected by the Natural Products Repository and archived in its Frederick facility.
MTL helps to screen and purify compounds from NCI’s Natural Products Repository, the world’s largest and most diverse repository of natural product extracts (derived from terrestrial, marine and microbial organisms).
A number of “new drugs that have been approved in the last 10 years are natural products or chemical derivatives from them,” said CCR director Robert Wiltrout. The program “is providing a tremendous chemical diversity that is not easily replicated by using synthesized small-molecule chemical libraries.”
To read an article on the Natural Products Repository, go to http://home.ccr.cancer.gov/connections/2008/Vol2_No2/news_6.asp
Recent Research Advances at NCI Labs in Frederick
- Biopharmaceutical Development Program (BDP) Supplies Antibody for Childhood Cancer Treatment: One of BDP’s early projects in the 1990s was producing monoclonal antibodies—when no pharmaceutical company would—to treat children with neuroblastoma, a common childhood cancer that grows in parts of the nervous system (http://www.cancer.gov/ncicancerbulletin/100510/page5).
- Nanotechnology in Reformulated Cancer Drugs: FNLCR’s nanotechnology researchers have worked with companies on nano-size compounds that show promise in destroying tumors.
- CCR researchers in Frederick helped develop an antibody named m102.4 that protected monkeys infected with the deadly Hendra virus and might be developed into a possible treatment for people.
- For more on recent advances, go to http://ncifrederick.cancer.gov/Science/ResearchOverview.aspx.
- A new function for the breast cancer gene BRCA1 was discovered by CCR researchers in Frederick: In addition to functioning as a tumor suppressor, it can also silence another gene that causes cancer when over-expressed.
- For more successes, go to http://ncifrederick.cancer.gov/Science/ResearchOverview.aspx.
Acknowledgements: The NIH Catalyst and the author would like to thank the following PEOPLE who contributed information and photos for this article: Andrea Frydl (NCI-Communications), Frank Blanchard (SAIC-Public Affairs), Brenda Boersma-Maland (NCI-CCR); AND KIMBERLY MARTIN (NCI-CCR)
For more information about the Frederick National Laboratory for Cancer Research, go to http://frederick.cancer.gov.
This page was last updated on Friday, April 29, 2022
