Monoclonal antibody prevents malaria in small NIH trial
One dose of a new monoclonal antibody discovered and developed at the National Institutes of Health safely prevented malaria for up to nine months in people who were exposed to the malaria parasite. The small, carefully monitored clinical trial is the first to demonstrate that a monoclonal antibody can prevent malaria in people. The trial was sponsored and conducted by scientists from the Vaccine Research Center (VRC) of the National Institute of Allergy and Infectious Diseases (NIAID), part of NIH, and was funded by NIAID. The findings were published today in The New England Journal of Medicine.
“Malaria continues to be a major cause of illness and death in many regions of the world, especially in infants and young children; therefore, new tools are needed to prevent this deadly disease,” said NIAID Director Anthony S. Fauci, M.D. “The results reported today suggest that a single infusion of a monoclonal antibody can protect people from malaria for at least 9 months. Additional research is needed, however, to confirm and extend this finding.”
According to the World Health Organization, an estimated 229 million cases of malaria occurred worldwide in 2019, resulting in an estimated 409,000 deaths, mostly in children in sub-Saharan Africa. So far, no licensed or experimental malaria vaccine that has completed Phase 3 testing provides more than 50% protection from the disease over the course of a year or longer.
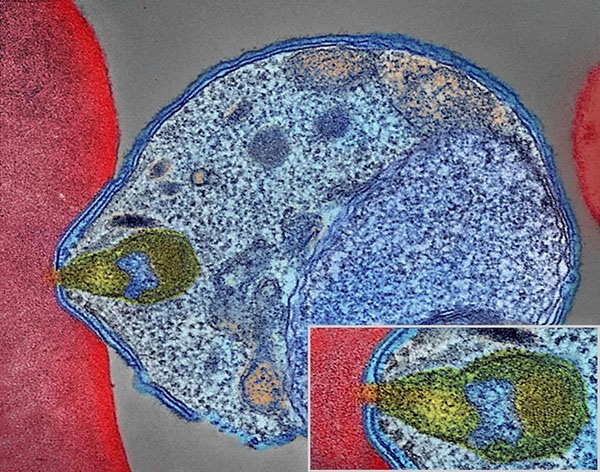
Colorized electron micrograph showing malaria parasite (right, blue) attaching to a human red blood cell. The inset shows a detail of the attachment point at higher magnification.
This page was last updated on Friday, January 21, 2022